Title of the experiment
Carboxylic Acids and Esters: Preparation of Methyl Salicylate (Oil of Wintergreen).
Objective of the Experiment
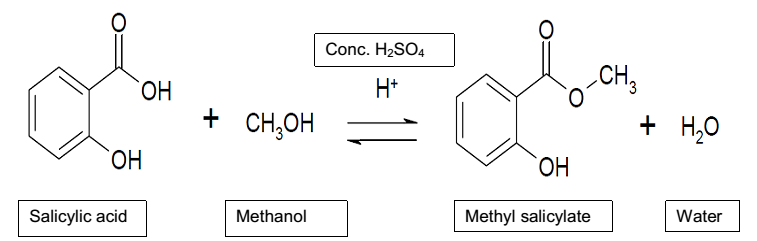
The objective of the experiment is to synthesize methyl salicylate, which is an ester, from salicylic acid and methanol through the process of esterification. The experiment achieves the objective by reacting salicylic acid and methanol in the presence of concentrated sulfuric acid and under the reflux condition to form methyl salicylate and water.
Synthetic Equation for the Reaction
Physical Properties of Reactants and Products
Salicylic Acid
Molar mass: 138.12 g/mol
Molecular formula: C7H6O3

Structure Boling point: 2110C
Melting point: 158-1610C
Density: 1.4 g/mL
Solubility in water: 1.8 g/L
Hazards: Highly flammable and harmful
(Royal Society of Chemistry par. 2)
Methanol
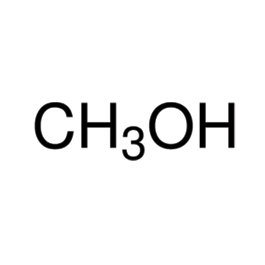
Structure
Molar mass: 32.04 g/mol
Molecular formula: CH3OH
Boling point: 65.40C
Melting point: -980C
Density: 0.791 g/mL
Solubility in water: Soluble
Hazards: Toxic, harmful, and highly flammable
(Royal Society of Chemistry par. 2)
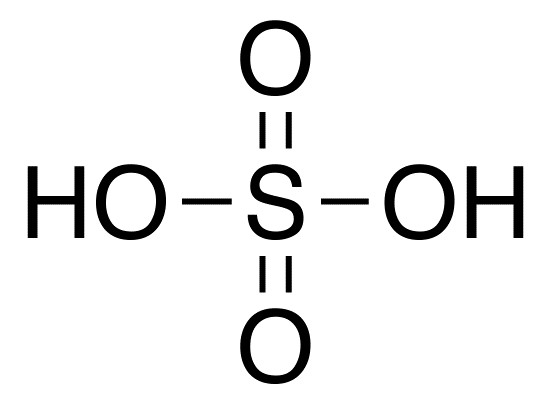
Sulfuric Acid
Molar mass: 98.08 g/mol
Molecular formula: H2SO4
Boling point: 2900C
Melting point: 100C
Density: 1.84 g/mL
Solubility in water: Soluble
Hazards: Corrosive, toxic, harmful, and highly flammable
(Royal Society of Chemistry par. 4)
Methylene Chloride
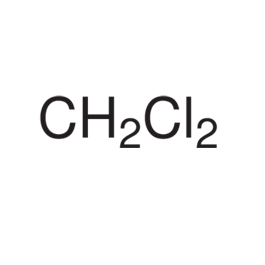
Structure Molar mass: 85.94 g/mol
Molecular formula: CH2Cl2
Boling point: 39.8-400C
Melting point: -970C
Density: 1.325 g/mL
Solubility in water: Soluble
Hazards: Harmful
(Royal Society of Chemistry par. 1)
Sodium bicarbonate
Structure Molar mass: 98.08 g/mol
Molecular formula: NaHCO3
Boling point: 8510C
Melting point: 3000C
Density: 2.16 g/mL
Solubility in water: 0.09 g/mL
Hazards: Irritant and harmful
(Royal Society of Chemistry par. 3)
Anhydrous Sodium Sulfate
Molar mass: 142.01 g/mol
Structure Molecular formula: Na2SO4
Boling point: 17000C
Melting point: 8840C
Density: 2.68 g/mL
Solubility in water: 18.5 mg/L
Hazards: Irritant
(Royal Society of Chemistry par. 2)
Methyl Salicylate
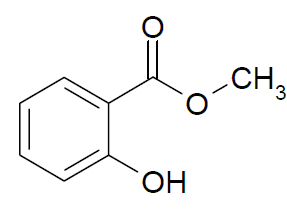
Structure Molar mass: 152.15 g/mol
Molecular formula: C8H8O3
Boling point: 2190C
Melting point: -50C
Density: 1.174 g/mL
Solubility in water: Soluble
Hazards: Harmful
(Royal Society of Chemistry par. 1)
Water
Molar mass: 18.02 g/mol
Molecular formula: H2O
Boling point: 1000C
Melting point: 00C
Density: 1 g/mL
Hazards: Corrosive
(Royal Society of Chemistry par. 4)
Procedure
The experiment was started by weighing 4.9 grams of salicylic acid and placing them in a 50 ml round-bottom flask. Then, 12.5 ml of methanol was measured and added to the 50 ml flask containing salicylic acid. The contents of the flask were swirled to ensure that methanol dissolved all the salicylic acid added. To catalyze the reaction, 5 ml of sulfuric acid was measured and added slowly to the flask while swirling cautiously. Subsequently, a reflux condenser was connected to the flask and the mixture was heated at reflux for 45 minutes for them to react. The reacting contents were swirled occasionally during the reflux period as the layer of oil gradually formed on top of the flask.
The reacting mixture was then cooled to room temperature after 45 minutes of heating at reflux. Before the contents were transferred to a separatory funnel, 10 ml of ice water was added to the contents. The ester formed, which is methyl salicylate, was extracted in a gentle manner with two portions of 15 ml methylene chloride. The two portions of methylene chloride extracts were combined and consecutively washed with 15 ml of water. Subsequently, the combined portions were washed with15 ml of 5% aqueous sodium bicarbonate solution. The organic layer containing methyl salicylate was dried over sodium sulfate and filtered into a pre-weighed 100 ml round-bottom flask using gravity filter. The solvent in the organic products was evaporated using a rotary evaporator. Given that the product is oil, evaporation was done cautiously to prevent its loss with the solvent. The product obtained in the flask was weighed and the percent yield calculated. Ultimately, IR and NMR were run to analyze and confirm if the product formed was methyl salicylate.
Calculation of the Theoretical Yield and Actual Yield
Determination of the limiting reactant:
- Moles of salicylic acid: 4.9g/138.12 g/mol= 0.035 moles
- Mass of methanol: 12.5 ml * 0.791 g/ml = 9.88 g
- Moles of methanol: 9.88 g/32.04 g/mol = 0.308 moles
- Hence, the calculations indicate that salicylic acid is the limiting reactant
Theoretical yield
- The esterification reaction shows that salicylic acid and methyl salicylate has a reaction ratio of 1:1.
- The ratio implies that 0.035 moles of salicylic acid forms 0.035 moles of methyl salicylate.
- Therefore, mass of methyl salicylate = moles × molar mass of methyl salicylate
- The mass of methyl salicylate = 0.035 × 152.15 g = 5.3 g
Actual yield
- Mass of the empty round flask = 52.72 g
- Mass of the round-bottom flask + product = 57.72 g
- Mass of the product= 57.72 g – 52.72 g = 5.00 g
Percent yield
- Percent yield of the product = Actual yield/Theoretical yield × 100
- Percent yield = 5.0 g/5.3 g × 100 = 94.33%
Synopsis of Results
The results indicate that esterification reaction occurred and led to the production of methyl salicylate. Calculation of moles shows that salicylic acid is the limiting reactant because it has a lower number of moles than methanol. Hence, using moles of the limiting reactant, theoretical yield of methyl salicylate is 5.3 g. Calculations also indicate that the actual yield of methyl salicylate is 5.00 g, which constitutes 94.33% of the theoretical yield. Such as a high percent yield shows that the esterification process was efficient. However, the percent yield was not 100% due to errors associated with the experimental setup. Thus, the possible sources of errors are an incomplete reaction of salicylic acid and methanol, evaporation of the product during reflux heating, and the loss of the product in the processes of extraction and washing.
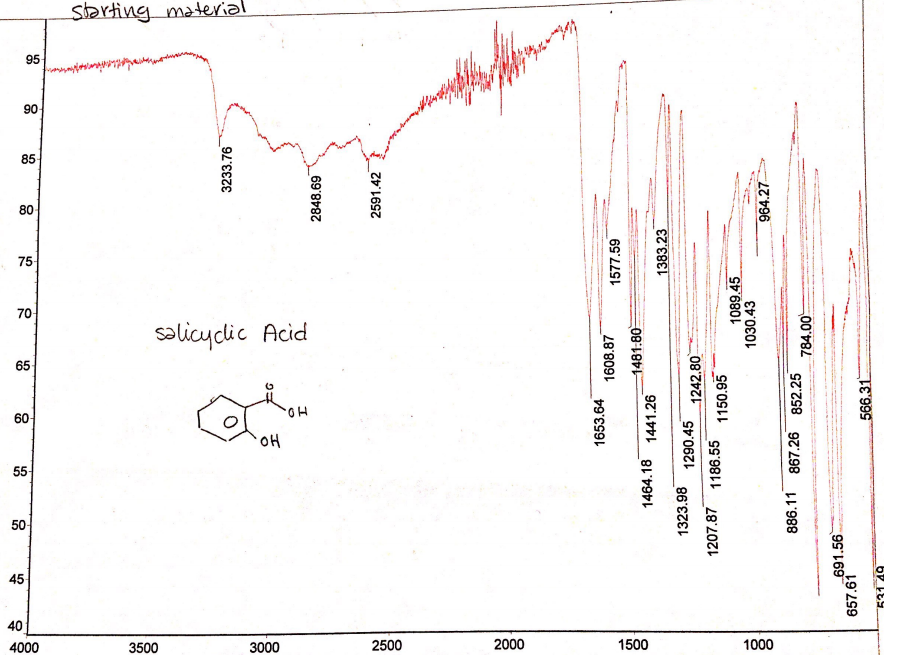
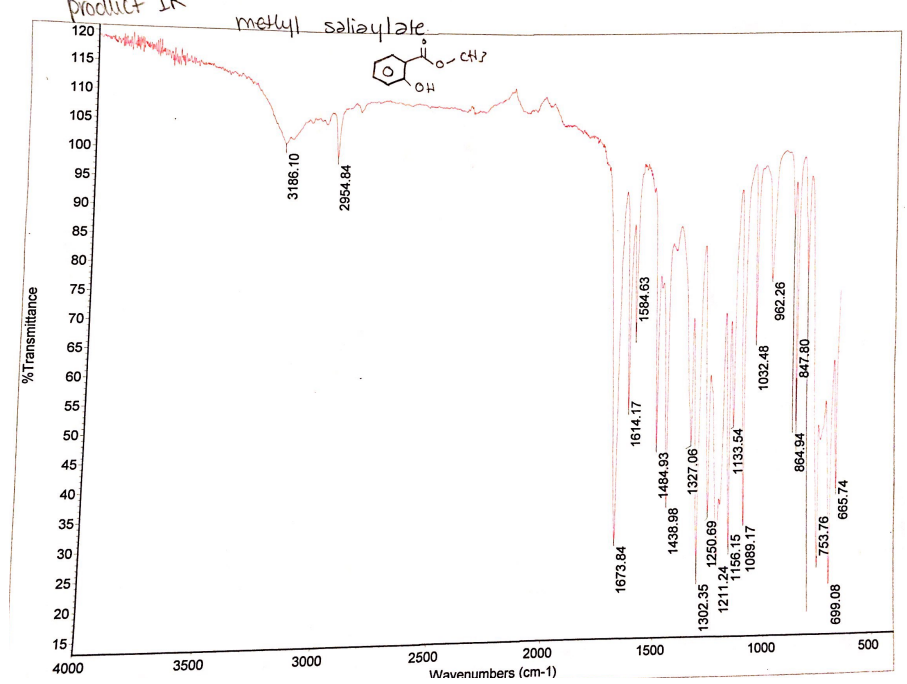
The IR spectra reveal that there is amarked difference between the IR spectra of the starting material and the IR spectra of the product. The starting material has apparent O-H stretches at 2848.69 cm-1, 2591.42 cm-1, and 3233.76 cm-1 (Figure 2). Comparatively, the IR spectra of the product have C=O stretch at 1673.77 cm-1 and O-H stretches at 3180.62 cm-1 and 2954.77 cm-1 as shown in Figure 3.
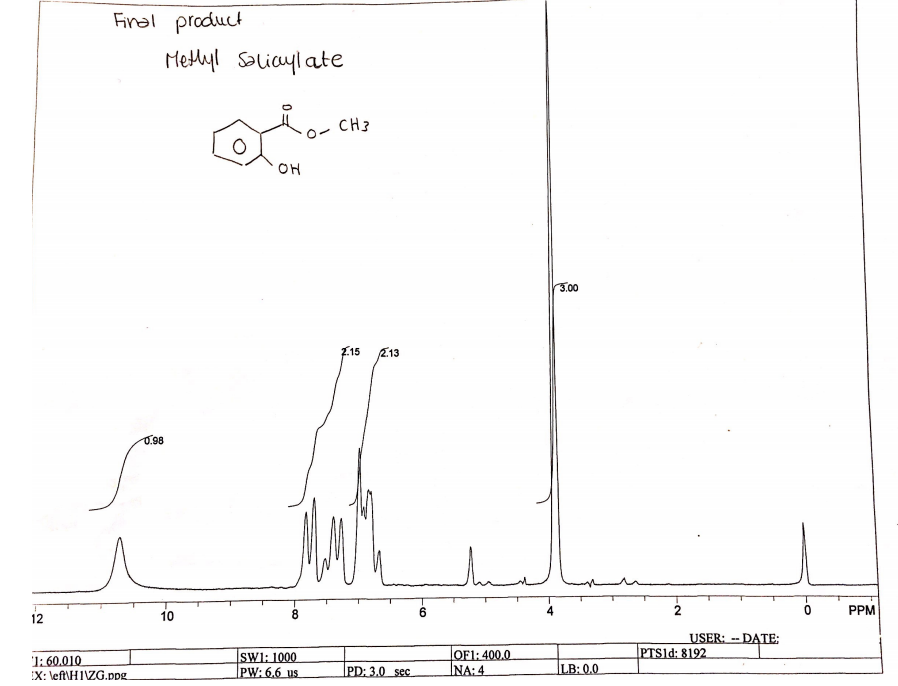
The pattern of chemical shifts in the NMR spectra of the product depicts the chemical shifts of methyl salicylate. The NMR spectra have four peaks of 0.98, 2.13, 2.15, and 3.0 (Figure 4). The peak of 0.98 is singlet peak at 11 PPM, the peaks at 2.13 and 2.15 are doublet peaks at between 6-8 PPM, and the major peak of 3.0 is singlet at 4 PPM.
Discussion
Synthesis of esters occurs through an important chemical reaction in organic chemistry called esterification. Essentially, esterification is an organic reaction that involves the reaction of carboxylic acid and alcohol in the presence of acids and heat. According to Whitmore, an ester bond forms between the alcohol and the carboxylic acid and there is the formation of water (37). In this case, salicylic acid and methanol reacted in the presence of concentrated sulfuric acid and under reflux conditions to form methyl salicylate and water. In the mechanism of esterification, concentrated sulfuric acid protonates carbonyl group of salicylic acid and creates a primary carbocation, which attracts the lone pairs of electrons in the oxygen species of the methanol. The formation of the carbocation and nucleophilic attack by methanol creates a resonance structure that allows the transfer of protons from the –OH group of methanol to the –OH group of salicylate. The transfer of hydrogen ion to the –OH group creates a good leaving group resulting in the loss of water molecule.
Concentrated sulfuric acid was used in the experiment because they create an acidic condition, which catalyzes the reaction between salicylic acid and methanol through protonation. As heat hastens the reaction rate and favors the formation of methyl salicylate, the reacting contents were heated under reflux condition for 45 minutes. Reflux system is a special technique of heating reacting chemicals to their boiling points for an extended period without causing their loss through the evaporation because it occurs in a closed system that prevents vapor from escaping (Schneider 19). The purpose of the reflux system is to increase the reaction rate and prevent the loss of reactants via evaporation.
The addition of ice water into the reacting contents forms an organic layer comprising the product and the aqueous layer comprising a solution of salts and acids. The position of the organic and aqueous layers varies according to their densities. Methyl salicylate is denser than water because their densities are 1.174 g/ml and 1 g/ml respectively. Hence, methyl salicylate is in the lower layer while the aqueous layer is in the upper layer.
In the extraction, methyl salicylate was extracted twice with two portions of 15 ml methylene chloride. The first extraction creates an equilibrium level between methylene chloride and the aqueous layer, which allows mass transfer of the organic molecules into the organic phase. The second extraction creates another equilibrium level, which allows mass transfer of the remaining organic molecules into the organic phase (Behrens and Datye 397). The resulting effect of double extraction is enhanced extraction when compared to a single big extraction.
Important chemicals used in the experiment are sodium bicarbonate and anhydrous sodium sulfate. The role of sodium bicarbonate was to neutralize acids that remained in the organic phase containing methyl salicylate. These acids are traces of sulfuric acid and salicylic acid that did not react with methanol. The addition of sodium bicarbonate caused fizzing because there was the production of carbon dioxide. Moreover, the experiment used anhydrous sodium sulfate in drying the organic product, methyl salicylate. Anhydrous sodium absorbs a lot of water from solids and stores them as the water of crystallization in its lattice structure.
The IR spectra of the reacting material and product indicate that there are marked differences in stretches formed. Apparently, the reacting material has broad O-H stretches that are evident at 2848.69 cm-1 and 2591.42 cm-1. These O-H stretches represent salicylic acid. Moreover, the reacting material has an O-H stretch at 3233.76 cm-1, which represent methanol. The spectra show that the reacting material has a carboxylic acid (salicylic acid) and alcohol (methanol). The IR spectra of the product have no O-H stretch at 2591.42 cm-1 of carboxylic acid, which means that all have been converted to methyl salicylate.. However, the IR spectra have maintained O-H stretches at 3180.62 cm-1 and 2954.77 cm-1showing that there are traces of methanol for they were in excess. The IR spectra of the product indicate that there is the emergence of C=O stretch at 1673.77 cm-1. This stretch reflects the presence of ester (methyl salicylate) because it has a C=O stretch representing carbonyl group (Chukanov 921). Thus, the IR spectra confirm that methyl salicylate was formed from the reaction of salicylic acid and methanol.
The NMR spectra depict chemical shifts of methyl salicylate because it has four peaks, which represent different protons in methyl salicylate. Diehl, Wawer, and Holzgrabe note that the peaks represent different types of protons in molecules (35). The major peak of 3.0 at 4 PPM represents the three protons in the methyl group while a minor peak of 0.98 at 11 PPM represents a single –OH attached to the aromatic ring. The double peaks between 6-8 PPM represent protons that are in the aromatic ring of methyl salicylate. In this view, the distribution of chemical shifts in the NMR spectra confirms that the product is methyl salicylate as expected.
The determination of the yield shows that the percent yield of methyl salicylate is 94.33% of the theoretical yield. The percent yield was not 100% because of the experimental errors, which cumulatively contributed to the loss of the yield. In this experiment, the incomplete reaction of salicylic acid and methanol is one possible source of the error that contributed to the loss of yields. Although the heating happened under reflux conditions, overheating of the reacting mixture might have caused the product to evaporate. As the experimental process involved steps of extraction and washing, these steps contributed to the loss of the product for they are not efficient.
Works Cited
Behrens, Malte, and Abhaya Datye. Catalysis for the Conversion of Biomass and Its Derivatives. New York: Cengage Learning. 2013. Print.
Diehl, Bernd, Iwona Wawer, and Ulrike Holzgrabe. NMR Spectroscopy in Pharmaceutical Analysis. Elsevier Science Limited, 2008. Print.
Chukanov, Nelson. Infrared Spectra of Mineral Species: Extended Library, 2014. Print.
Royal Society of Chemistry. ChemSpider: Search and share chemistry. 2015. Web.
Schneider, Frank. Qualitative Organic Microanalysis: Cognition and Recognition of Carbon Compounds. Vienna: Springer Vienna, 2012. Print.
Whitmore, Frank. Organic Chemistry, Volume One: Part I: Aliphatic Compounds Part Ii: Alicyclic Compounds. Newburyport: Dover Publications, 2012. Print.