Abstract
Living organisms require water for various physiological processes such as respiration and transport of substances. Osmosis is the main physiological process through which water gets into living cells. Germination, which initiates the development of plants, is highly dependent on the availability of water. This experiment aimed at investigating the effect of various concentrations of KCl on the germination of mung bean seeds. Ten mung bean seeds were incubated in KCl solutions whose concentrations ranged from 0.1M to 0.5M for seven days during which the rate of germination was checked. It was observed that increasing the concentration of KCl led a decrease in the rate of germination of mung bean seeds. The study concluded that salts hampered the germination of seeds by inducing osmotic stress.
Introduction
Water is a crucial requirement for all living things because it works as a medium for numerous biochemical reactions (Golzardi et al. 2012). Living organisms take in water through physiological processes such as diffusion and osmosis. Osmosis can be illustrated as the advancement of water across a partially porous membrane from a zone of high water concentration to an area of low water concentration. For osmosis to go on, a dilution gradient has to be present between the two sides of a partially permeable membrane. One section of the membrane usually consists of the cell, whereas the other side of the membrane comprises the cell environment. Based on these differences, the solutions surrounding the cells can be described as hypertonic, isotonic or hypotonic.
The differences in the concentrations of these solutions determine the direction of water movement. A hypotonic solution contains more water molecules in comparison with the cell medium. Putting a plant cell in a hypotonic solution causes the cell to take in water and swell. An isotonic solution has a water potential that is equivalent to that of the cell. Therefore, placing a plant cell in such a medium leads to zero net movement of water, and the cell remains unchanged. A hypertonic solution, conversely, contains fewer water molecules than the cell. Therefore, a plant cell placed in this solution loses water to the surroundings. Consequently, the cell volume reduces, and the cell becomes limp.
Osmosis is particularly important in the initial stages in the life of plants, which normally begins with germination. Germination is delineated as the process that enables plants to develop from seeds. A number of attributes such as light, water and warmth are required for germination. Before germination, seeds are found in a state of dormancy meaning that the surrounding conditions do not favor the germination of the seed. Plant seeds take in water through the process of osmosis.
Water triggers germination through two key mechanisms. The first mechanism is through the facilitation of the hydrolysis of stored food substances in the endosperm. The hydrolyzed food substances are further broken down to yield energy that is used to drive the process of germination. In addition, water softens the seed coat (testa) thereby making it easier for the newly formed roots and shoots to emerge through the micropyle. The second mechanism is through activation of hormones that stimulate growth (Kim et al. 2008). Therefore, water is essential in the germination process.
Different environmental conditions limit the availability of water to plants thereby hindering the propagation of seeds. Such conditions include drought and salinity (Guo et al. 2012). During drought, there is inadequate water in the soil, which makes it hard for the plant to obtain sufficient water. Conversely, in saline conditions, water is available though it contains a mixture of salts that make the soil hypertonic with regard to the plant cells.
This experiment intended to establish the effects of diverse salt strengths on the germination of seeds. Potassium chloride was the main salt used in the experiment. It was predicted that increasing the concentration of KCl in solutions would lead to a decrease in the rate of germination.
Materials and Methods
In this experiment, effect of salts on germination was investigated using 20 mung bean seeds. The test solution was 12 ml of 0.25M KCl solution while the control was 12 ml of deionized water. Ten mung bean seeds were placed in two paper towels, which were folded twice. The paper towels were then moistened using 12 ml of deionized water. A similar setup was made for the other ten mung bean seeds, which were moistened with 12 ml of the experimental solution (0.25M KCl).
Thereafter, each group of ten seeds was covered with towels and put in separate Ziploc bags. The two bags were then kept near a well-lit window at room temperature (between 20°C and 23.5°C). The seeds were checked every day after 3 pm EST to monitor the germination. Observations were made for seven consecutive days. This experiment was also performed to check the effect of different concentrations of KCl (0.1M, 0.4M and 0.5M) on germination. The number of seeds that germinated was counted and recorded.
Results
The rate of germination was tested using deionized water (control) and different concentrations of KCl (0.1M, 0.25M, 0.4M, and 0.5M). The percentage germination of all the seeds in the control and KCl solutions was zero on the first day. However, the seeds in deionized water began germinating on the second day. The percentage germination of these seeds was 41.1% on the second day, which increased to 83.3% and 95.6% on the third and fourth days respectively.
By the end of the fifth day, all seeds had germinated. The seeds placed in 0.1M KCl began germinating on the second day at a rate that was comparable to that of the seeds in deionized water. However, the complete germination of the seeds was attained at the end of the seventh day while in deionized water complete germination was attained at the end of the fifth day (table 1).
Table 1: The percentage germination of mung bean seeds in different concentrations of KCl over a seven-day period.
The seeds in the KCl 0.25M started germinating on the second day. However, the rate of germination was considerably lower (2%) than the rate of germination in deionized water and 0.1M KCl. In addition, there was only 73.3% germination by the end of the seventh day. In 0.4M KCl, the germination of mung bean seeds began on the third day reaching a maximum percentage of 32.5% by the fifth day. The 0.5M KCl solution produced the lowest rate of germination with initial germination being observed on the sixth day at a rate of 1.67%. The highest rate of germination for this solution was 3.33% at the end of the seventh day.
A graphical representation of the above information was provided in figure 1.
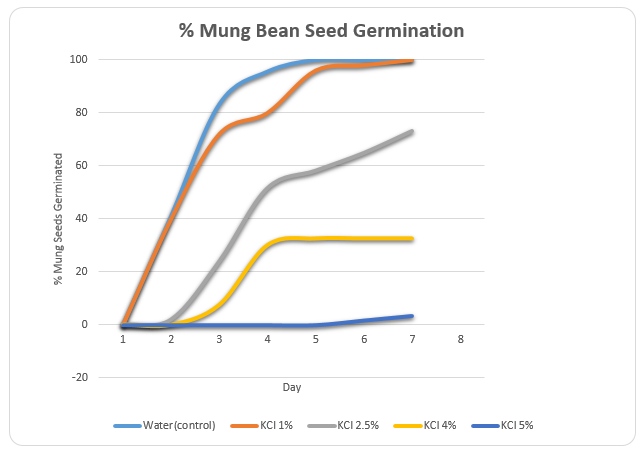
It was shown that deionized water had the highest rate of germination compared to the KCl solutions. Of the KCl solutions, it was noted that 0.1M KCl had the highest germination rate while 0.5M solution had the lowest rate of germination. The overall trend witnessed was that the rate of germination reduced as the concentration of KCl increased.
Discussion
This practical aimed at investigating the effect of various strengths of KCl on the germination of mung bean seeds. It was observed that the seeds in deionized water sprouted at a higher rate compared to those placed in KCl. It was also noted that the rate of germination decreased as the concentration of KCl rose. The results of this experiment supported the prediction that increasing the concentration of KCl would lead to a decrease in the rate of germination. It was evident from the percentage germination KCl solutions that an increase in the concentration of KCl translated to a decrease in the germination rate.
The above observations could be attributed to the effect of osmosis on plant cells. The seeds in deionized water were able to imbibe water via osmosis because the deionized water was hypotonic in relation to the seeds. Water molecules, therefore, moved from the paper towel into the cell. The water then triggered the hydrolytic enzymes that hydrolyzed stored food within the seed thereby yielding energy that was a prerequisite for germination.
On the other hand, there were low rates of germination of the seeds in KCl solutions. However, the rate of germination was higher in 0.1M KCl solution compared to the other concentrations. This observation was because the low concentration of KCl in the 0.1M solution did not affect the concentration gradient adversely. Therefore, water was able to move from the 0.1M KCl solution into the seeds leading to the germination of the seeds.
However, the rate of germination was still lower than that attained in deionized water. In contrast, the mung bean seeds placed in 0.5M KCl solution did not germinate because osmosis was hampered. The 0.5M solution was hypertonic with respect to the bean seeds. However, the water did not move out of the seeds into the solution because the seeds were dry due to their state of dormancy. There was no movement of water into the seeds. Consequently, seed dormancy was not broken, and germination was unable to proceed.
The findings of this study were in agreement with other similar studies, which showed that salts prevented the germination of seeds. For example, a study by Brandenburg and Kleier (2011) showed that MgCl2 at a concentration of 0.1M lowered the rate of germination of radish. However, in this study, the researchers used 0.01M MgCl2 as the control instead of deionized water. A separate study by Sholi (2012) also indicated that salt concentrations had adverse effects on germination and development of tomato cultivars (monocotyledons).
Liu (2012) also showed that increasing the concentrations of NaCl and sorbitol lowered the rate of germination due an elevated expression of ZAT6 in leaves. All these studies showed that salt concentrations affected the germination of dicotyledons and monocotyledons equally.
Conclusion
Increasing the concentration of KCl impeded osmosis and led to low rates of germination. The findings of the experiment led to the deduction that water was essential for germination and that saline conditions had injurious upshots on the rate of seed germination.
Works Cited
Brandenburg, William, and Catherine Kleier. “Effect of MgCl2 on Germination, Growth and Biomass Allocation of the Radish CV. “Cherry Belle”.” American Journal of Environmental Sciences 7.2(2011): 132-135. Print.
Golzardi, Farid, Saeed Vazan, Hossien Moosavinia and Ghasem Tohidloo. “Effects of Salt and Drought Stresses on Germination and Seedling Growth of Swallow Wort (Cynanchum acutum L.).” Research Journal of Applied Sciences, Engineering and Technology 4.21(2012): 4524-4529. Print.
Guo, Youyan Wenhui Zhang, Jingfeng He, Jianyun Zhou, and Hongyuan Yu. “Effects of water stress and seed mass on germination and antioxidative enzymes of Xanthoceras sorbifolia.” African Journal of Biotechnology 11.18(2012): 4187-4195. Print.
Kim, Sang-Gyu, An-Kyo Lee, Hye-Kyung Yoon, and Chung-Mo Park. “A Membrane-Bound NAC Transcription Factor NTL8 Regulates Gibberellic Acid-Mediated Salt Signaling in Arabidopsis Seed Germination.” The Plant Journal 55.1 (2008): 77-88. Print.
Liu, Xiao-Min. “Phosphorylation of the Zinc Finger Transcriptional Regulator ZAT6 by MPK6 Regulates Arabidopsis Seed Germination under Salt and Osmotic Stress.” Biochemical and Biophysical Research Communications 430.3 (2013): 1054–1059. Print.
Sholi, Nasser, J. Y. “Effect of salt stress on seed germination, plant growth, photosynthesis and ion accumulation of four tomato cultivars.” American Journal of Plant Physiology 7.6 (2012): 269-275. Print.