Introduction
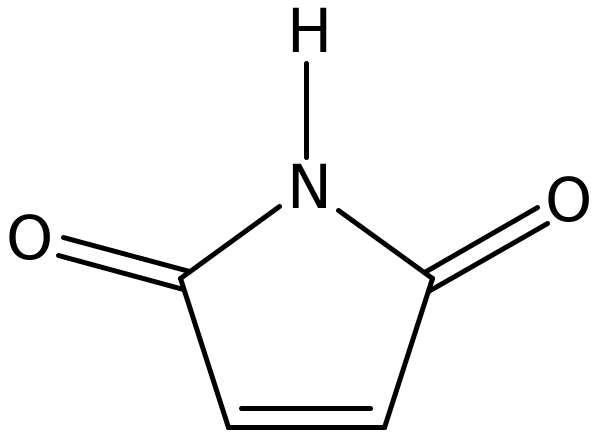
Maleimide is unsaturated imide which is commonly used as a building block in organic synthesis. The general chemical formula of this compound is H2C2(CO)2NH. The IUPAC name of maleimide is 2,5-Pyrroledione. Its molecular formula is C4H3NO2 with a molar mass of 97.07 g/mol and a melting point 91-93 °C (Hiran et al. 2007, p. 386).
Some Maleimides compounds occur naturally and are evidently oxidation products of tetrapyrrole compounds in sediments, although the nature and time of the oxidation have not yet been clarified. Recent developments in research have focused on the industrial production of maleimides due to their increased use in a wide variety of industries. Some of the examples of maleimides compounds include;
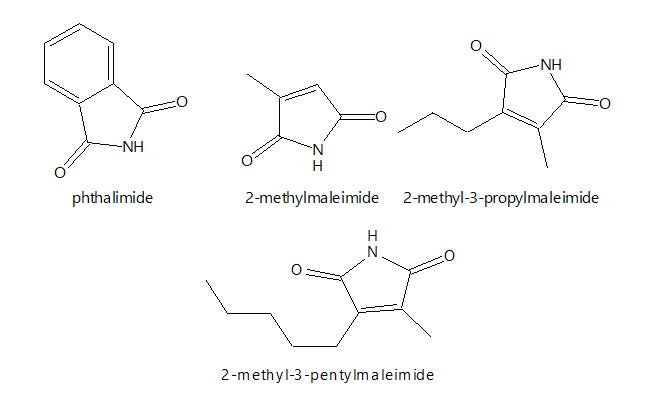
Maleimides are soluble in most organic solvents such as tetrahydrofuran (THF), dimethylformamide (DMF),dimemethylsulfoxide (DMSO), ethyl acetate, acetone, carbon tetrachloride, chloroform, dichloromethane and cyclohexanone. The name, maleimide is formed from the names maleic acid and the functional imide group -C(O)NHC(O). Typically, maleimides derivatives are formed when the NH group is replaced by either aryl or alkyl groups.
Such groups can constitute methyl, ethyl or phenyl group. The NH group in maleimides is mostly attached to a long chain group either aliphatic or aromatic groups to form a polymer such as polyethylene glycol.
The blood substitute referred to as MP4 constitutes a chemical combination of haemoglobin with maleimide-polyethylene glycol. Maleimides compounds possess a vast of good physical characteristics which has contributed to their wide use in most chemical and industrial fields. For instance, they are thermally stable compared to other monomers like vinyl compounds and therefore they are employed in the manufacturing of synthetic resins. Such resins (polymers) modified by maleimide become heat resistance. Maleimide also has a reactive site-selective characteristic enabling its common application in the drug industry and biotechnology where they link the proteins and the surfaces through a flexible attachment (Simionescu & Grigoros, 1990, p. 43).
Synthesis of maleimide compound
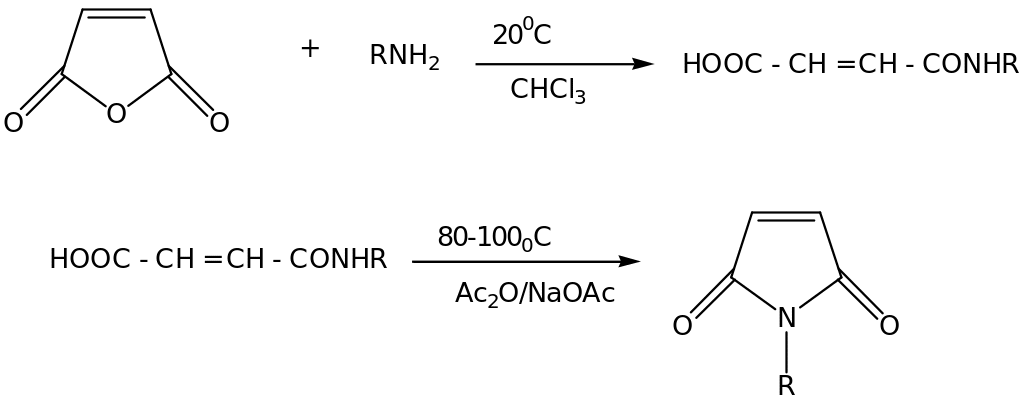
The Synthesis of N-arylmaleimides (RMI) proceeds via two-step mechanism. In the first step, arylamines (RNH2) react with maleic anhydride (MAN) to form maleamic acid, RNHCOCH=CHCOOH. The second step cyclization occurs by heating in acetic anhydride solution in the presence of anhydrous NaOAc under elimination of water (Hiran et al. 2007, p. 386).
The kinetics and thermal properties
The N-substituted maleimides form an important class of electron acceptor monomers that are capable to form charge transfer complexes with a wide variety of vinyl monomers. Copolymers are formed by the free radicals maleimides with electron donors, a process referred to as copolymerization. Besides their general application in the preparation of a range of polymers and copolymers, the N-substituted maleimides are important biologically active molecules.
For example, the N-ethymaleimide is a diuretic and an important nephrotoxic agent in dogs. Reactions involving maleimides compounds may take place in either basic or acidic medium. It has however been observed that at pH of 7, the reaction becomes highly selective. For instance, the reactivity of sulfhydryl groups is a thousand times faster than the one involving the amine group. Copolymerization of maleimides has provided a method of preparing highly thermal stable polymers. In addition, incorporating more flexible units within the polymer backbone can enhance the processability of maleimide polymer.
There are, however, several draw backs during processing, for example, maleimide polymers in fully imidized form are unmanageable and various difficulties in processing due to insolubility. The copolymerization process has resulted in improved polymers with unique characteristics and properties that meet specific requirements hence there popular use in industries. Most copolymers of maleimides compounds have demonstrated as having better thermal stability than the polymers made with vinyl monomers subunits. Thermal properties of copolymers and polymers are investigated using thermo-gravimetric analysis (TGA).
Polymaleimides are therefore potential heat and chemical resistance compounds (Chunqing, Huaming & Pengsheng 2004, p. 1161). The recent major developments in biotechnology areas namely, therapeutic proteins and other biomolecules such as the antibodies as well as the antibodies fragments have drawn researchers to study more on maleimides compounds. These biotechnology products are nowadays prepared on a large scale basis making them readily available in the market.
The use of these products is becoming limited as several disadvantages are associated with their physical and chemical characteristics. The antibodies are very unstable and they rapidly degrade into fragments. In addition, their absorption or digestion degree in the body is low. These biotechnology products are highly administered especially in pharmaceuticals areas in the manufacture of drugs and therefore various ways have been developed to overcome this set back.
To increase the biological activity of therapeutic molecules in the manufacture of drugs, proteins are covalently bonded to polyethylene glycol which is a soluble in water. Recent studies on the biological properties of PEG – modified proteins (PEG – conjugates or pegylated) proteins have shown considerable improvement compared to PEG-conjugates or pegylated proteins biological and physical properties. The reactive NH group in the maleimides compounds tends to photo-initiate reactions and is commonly employed in the industrial manufacture of polymers from ethylene unsaturated compounds such as the acrylate compounds. They are the most widely used monomers in the manufacture of polymer compound since they result in considerable heat resistance and high stable copolymers.
Stabilizing the maleimide compounds at room temperature, maleimide are stable compound and may occur as solid or liquid form. The storage is important because this compound tends to discolour to brown or blue blackish hence loosing its value. The solid maleimide is packed in bags and containers avoiding any contact with the moisture. The liquid maleimide is usually stored in tanks. Generally, the rate of discolouration of the compound will depend on the form, state and storage conditions of the stored maleimide. In addition, discolouration of the stored compound is inevitable although it could occur slowly or quickly.
Discolouration is a serious set back in the production and use of maleimide. Using maleimide compounds in the manufacture of thermoplastic resins to improve their heat resistance is facing a major draw back as the maleimide causes the vinyl chloride resins and the methyl methacrylate resins to discolour. This however lowers the quality of the product. As a result the commercial value of the thermoplastic resins decreases drastically.
It is there noteworthy to provide a stabilized form of maleimide compound that is difficult to discolour. Such a compound can be obtained by qualitative and quantitative preparation which will improve the storage stability and reduce the discolouration. The contents the primary amine and the maleic anhydride should be measured. For instance, using primary amine > 500 ppm and the maleic acid of 10 to 1000 ppm can lower discolouration considerably. The storage stability could also be enhanced by use of amines content of > 500 parts per million and 2-amino-N-substituted succinimide compounds content > 300 ppm.
These two methods are effectively employed when an aromtic group is used as the side chain (Sharma, & Husain, 1997, p. 781). Discolouration of the maleimide compound is caused by the presence of amines which is used as a raw material for synthesizing the compound and a considerable amount of chlorine compound contained in the maleimide compound. Organic solvents employed during the maleimide preparation are volatile and on evaporation process in the manufacture of resins, the primary amines and chloride ions are thickly concentrated on the surface of resins. This adds to the discolouration problem.
The maleimide compound with are stable and can be stored without affecting their desirable quality are prepared by qualitatively controlling the weight of each raw material within a specific range. There are a wide variety of maleimides prepared by this method of improving stability foe example, N-benzyl maleimides, N-cycloalkyl, N-methoxyphenyl maleimides among others. 1.2.2 Cross-linking process cross-linking is a terminology used in most industrial application that refers to the process of covalently joining two or more molecules. The molecules being used as cross-linkers must contain specific functional groups that are reactive.
Commonly used cross-linkers are the amines and sulfhydryls groups. These cross-linking reagents have been used in the determination of proteins, DNA, nucleic acids and molecules relationship in the cell membrane. In addition, cross-carriers are used in the solid-phase immobilization, hapten carrier conjugation; antibody-enzyme conjugates preparation, modification of drugs and solid surfaces. Cross-linking of molecules helps in the analysis of protein conformational that occurs before and after the interaction process. Depending on the type of the interacting molecule, different distances may result and are determined by typical comparison of cross-linkers using varying arm lengths. The conjugation of amines and sulfhydryls on a protein will furthermore show where the tertiary or the quaternary amino acid is situated along the protein domain.
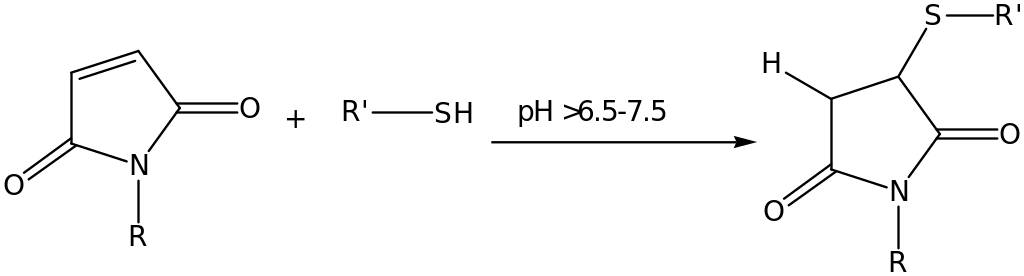
This is important since it exposes the structural composition of the protein. Other cross-linkers commonly used are the carbonyls, aldehyde group and carboxylic acids functional group. Mostly cross-linking process is selective. This is not always the case as shown in an examination of phenyl azide cross-linker. Maleimide is a reactive group, which is usually coupled with the functional group – sulfhydryl.
Further examination of this kind of attachment is that, the reactive sites used are cleaved and forms a number of more reactive sites increasing coupling. The reaction of maleimide compound with sulfhydryl groups takes place in a pH of 6.5 – 7.5 and a stable irreversible thioether compound is produced. Maleimides group have been experimentally observed to react 1,000-fold faster with sulfhydryls compared to amines, when the pH is >8.5 the reaction with primary amines is most favourable. Although maleimide compounds is known for its reactivity, reaction with histidines and methionines groups have failed in all the experiments conducted in the past two decades.
In basic pH medium, a considerable hydrolysis of the maleamic acid occurs to form reactive maleimides group. To avoid competitive reaction between the maleamic acid and thiol group, which are added in the reaction to react with excess maleimide, should be removed from the reaction. During the reaction, EDTA is usually added in the coupling buffer to avoid oxidation of the sulfhydryls. The following is the reaction mechanism of maleimide compounds and sulfhydryls group;
Maleimide derivatived surfaces In the immunological assays studies, surfaces are generally attached to protein by maleimide compounds. The reactive sulphydryl groups are covalently bonded to maleimide groups which are attached on the surface. The derivatized surface is prepared by the introduction of SH terminus leading to the reduction of disulphide bonds (Oishi, Kagawa & Fujimoto, 1993, p. 27). 1.2.3 Reduction of disulphide bonds The disulphide bonds formed between two cysteine molecules (shown below) is highly stable making the biomolecules being coupled non-reactive.
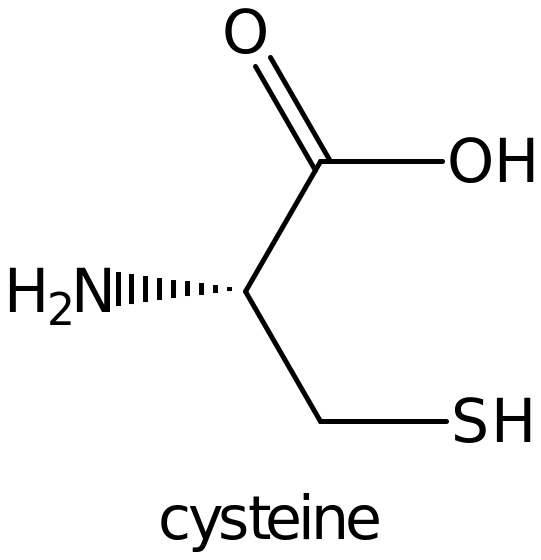
Reduction of disulphide groups involves the conversion of oxidized form to the reduced form generating two sulphydryl groups. Reducing agents used in this experiment include; the 2-Mercaptoethanol, S-Acetylmercaptosuccinic anhydride among others. These sulphydryl groups react readily with the Maleimide compounds on the plastic surface leading to a very stable covalent bond. The steps for the above reaction are shown in the reaction scheme below;
![]()
The amino group can also be transformed to sulphydryl group. The primary amines on amino acid Lysine or those on the terminal end of a biomolecule can further be modified by introducing the SH group. The sulphydryl group react with the maleimide compounds which are covalently bonded on the surface. The following reaction scheme illustrates the preparation of the biomolecule being attached to SH group through the Traut’s reagent (AR-S). The reaction can also occur by the use of 2-Iminothiolane hydrochloride as the reagent.
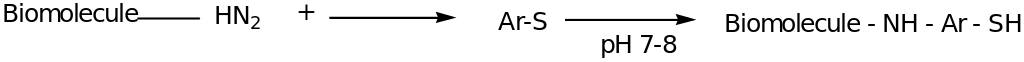
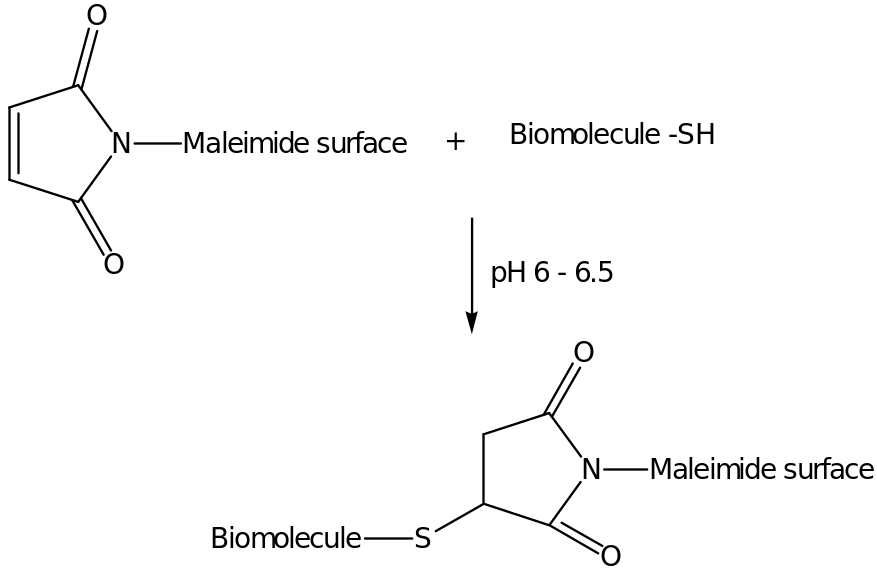
In the above reaction, EDTA is added to prevent oxidation of sulfhydryl groups reconstituting to form irreversible disulphide bonds. The attachment of the biomolecule-SH to the maleimide derivative is linked by the sulphydryl groups under the pH of about 6.5.
Principles of Maleimides synthesis with different amines
Maleimides compounds are typically prepared using direct amination of maleic anhydride and primary amines that produce maleamic acid intermediate. This is followed by dehydration process of the imides that results to cyclization using the acetic acid as the dehydrating agent. In this method, one mole of maleimide requires one mole of acetic anhydride to produce two mole of acetic acid. This, however, causes corrosion in the equipment and results in consequence treatments and pollution in the environment. Different methods have been proposed in the production of maleimides. Maleimides compounds can be prepared using the solid catalyst of acidic nature rather than the liquid-acidic form. This method was patented in the United State Patent No. 2,444,536. Sodium acetate and acetic anhydride are employed in the reaction as catalysts.
The main method of maleimide synthesis may also occur when Maleic anhydride is reacts with primary amine in presence of a solid-acidic catalyst. The reaction is heated to around the temperature of about 100-1800C. In completion of the reaction, the solid catalyst and the crude maleimide are separated (Oishi & Fujimoto, 1992, p. 1187). To obtain pure maleimide, the crude product is subjected to two step techniques. The first one is extraction followed by crystallization techniques.
The primary amine is usually characterized by either straight chain containing cyclic rings or aromatic substituents. Most primary ions are composed of 2-10 carbon atoms. The most preferred organic solvents are the methylbenzene and the dimethylbenzenes with the polarity of 0.2. Two principles of preparing maleimide are being investigated. One involve the use of n-hexane organic solvents with the polarity of less than 0.05 and the second one is the use of have dimethylbenzenes with the polarity of 0.2. Commonly used solid catalysts are inorganic acid supported by a neutral diatomite or silica gel carrier. For this reaction, strong acid are preferred such as the phosphoric acid or sulphuric acid (inorganic acid).
The solid acidic catalyst is prepared by heating to about 200-4000C and constitutes about 50 to 200 percent relative weight of maleamic acid. The maleimide reaction takes place in a low-polarity organic solvent with a boiling solvent of about 100 and 2500C. On completion of the reaction, the solid- acidic catalyst and the solvent mixture are separated by filtration technique. The solid catalyst is then recycled and the crude maleimide obtained by evaporating the solvent. Pure maleimide compound is therein obtained by fractional distillation of the crude maleimide compound under compressed pressure.
The distilled product is thermally unstable and degrades immediately after formation. When the two-step of extraction and crystallization are followed, the maleimide reaches a purity greater than 99.5 wt %. This method has an important advantage in that it can use both batch and continuous reactors for high yield products. Separation of the two reactants, the catalyst and the product is easily manageable since the reaction takes place in two different phases namely, the solid and the liquid phase. Thus the two are separated at room temperature. However, separation could also occur and different temperature and still the recycled catalyst maintain its selectivity and activity properties. Another method of synthesizing oligmers containing maleimide group is disclosed in the Japanese Patent No. 54-30155.
In this method, the quaternary amine salt of either organic or inorganic acids is used as the catalysts. Satisfactory activity and selectivity of the catalyst is high when a certain ratio of the salt and the acid is used. This means that the recycled catalysts must be modified after purification to attain the required ratio. This disadvantage is complexity and higher cost while using this method. 2.2 Maleimides synthesis under acidic condition
According to the disclosed UK Patent No. 1,041,027 and the United State Patent No. 3,431,276, maleimides compounds can be prepared using the azeotropic distillation system. Here, the catalyst is acidic in nature and is mixed with the volatile organic solvent. The reaction mixture is the heated to about 1000C and during which evaporation occurs to remove water. The organic solvents used in this reaction are, methylbenzene, dimethylbenzenes or chlorobenzenes. This method, however, requires a small amount of the expensive dehydrating phosphoric acid that renders the reaction unsatisfactory hence the process becomes uneconomical.
This problem can be solved in accordance with the Japanese Patent Publication No. 53-68700 and Japanese Patent Publication No. 57-42043. The two patents disclose ways of increasing the yield of maleimide derivatives. According to the former patent, the problem can be overcome by adding aprotic polar solvent. This improves the solubility of the maleimide and substantially increases the yield.
The use aprotic polar solvent is however hampered by its high boiling point properties as becomes difficult to remove from the solution. Further, a solution containing the non-polar solvent, polar solvent and the amino acids catalysts such as o-benzosulfonic acid have been examined and shown to improve the maleamic acid solubility in the reaction with maleic anhydride. In addition, this reaction is costly as the separation of the catalyst and the crude product is difficulty due to the fact that the reaction is in the polar phase (Oishi, Kamori & Fujimoto, 1992, p. 236).
For example, a pot-procedure which is typically used to synthesis maleimides derivatives with either the symmetrical or the unsymmetrical bis(heteroaryl) group is described in the Suzuki-Miyaura cross-coupling sequence. The reaction basically utilizes the use of the compounds namely; 3,4-diiodo-1-benzyl-1H pyrrole-2,5-dione with cyclic esters using PdCl2 (dppf) as the catalyst. The reaction is illustrated in the following scheme;
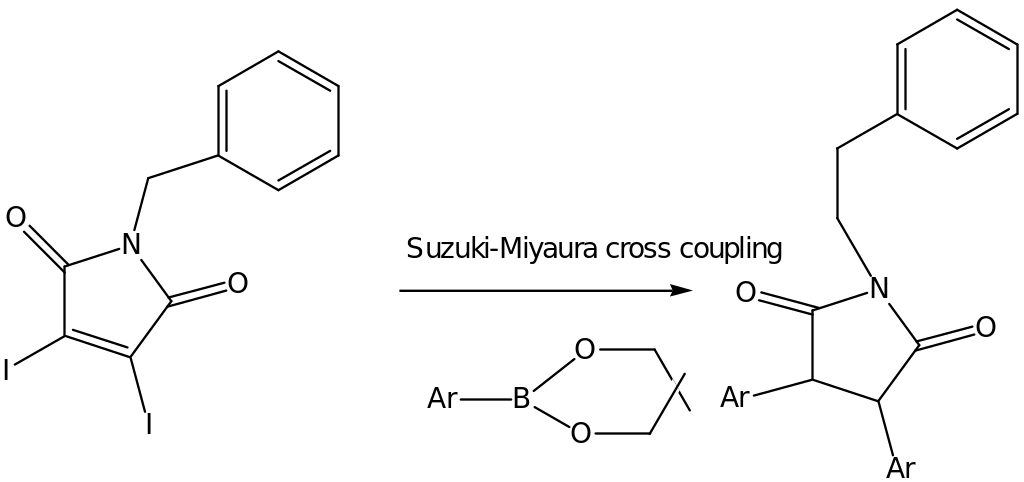
Another example is the ayrl-substituted maleimides which was produced and its activity against calmodulin dependant kinase determined. Other activities that were determined included the inhibitory whereby the enzyme ranged from 10 nm to 20 nm.
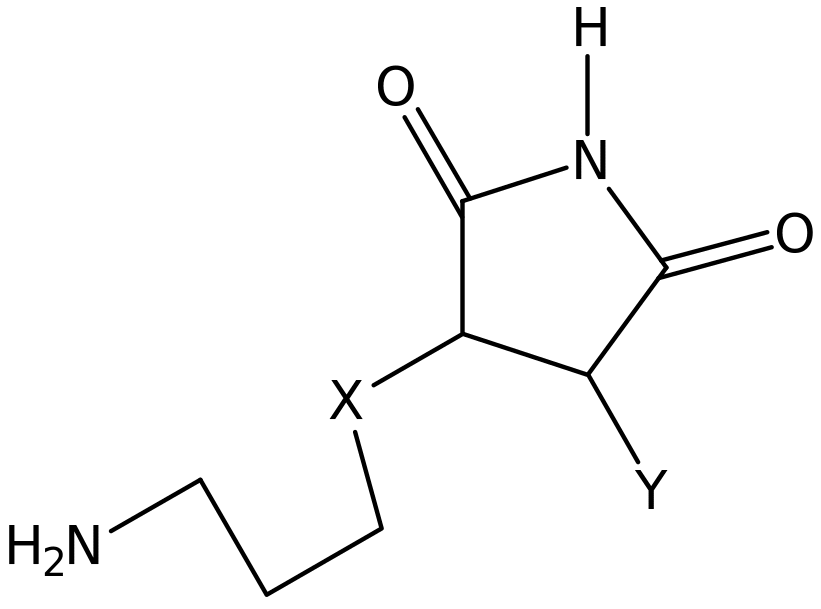
Maleimides synthesis using acetic anhydride
Classes of polymers that contain repeated imide units are referred to as polyimides. Polyimides are characterized by stable thermal properties like temperature resistance and being chemical resistant. Maleimides derivatives are a subclass of imides that contain olefinic functional group (alkene) that is used in the formation of polymers. Generally, synthesize of maleimide starts with the initial chemical reaction of the primary amine compound and the maleic anhydride resulting to maleamic acid which is followed by the ring-closure of maleamic acid. The ring-closure process is not achieved easily especially when the substituent chain that is attached to the primary amine is straight. Few methods have so far been reported to accomplish the ring closure step though none has been fully satisfactory.
Ring-closure is effectively established by the reaction between the maleamic acid, acetic anhydride plus sodium acetate which acts as catalyst. This method of preparing aromatic maleimides using acetic anhydride works well and purification is done by recrystalization technique. Addition work on preparation of aliphatic maleimide using this method has failed considerably resulting in low yield and purity of the desired product.
The resulting product is darkish in colour and this reduces the quality of the maleimide derivatives. Another method that has been used to complete the second step of closing the ring is by heating the maleamic acid dissolved in the azeotropic solvent under a solid acid catalyst. This reaction is accomplished by adding aprotic solvent which increases the reaction rate. However a great draw back of this method is that polar solvent are relatively expensive and the difficulty involved in the removing them after the reactions. As mentioned above aprotic solvents have several disadvantage particularly there toxic nature.
A number of maleimides compounds prepared comprising the N-propionitrile groups are formed by the reaction between the N-propionitrile-maleamic acid and acetic anhydride. These novel maleimides may be homo-polymerized or copolymerized using one or more comonomers. The United State Patent No. 2,444,536 further discloses the process for the synthesis of N-aryl maleimides. This reaction of aryl maleamic acid and acetic anhydride is heated with sodium acetate at about 100° C. The reaction is completed by addition of excess acetic acid (Wang et al. 2006, p. 940).
The reaction of the acid anhydride and the maleamic acid occurs effectively at mild condition. Heating is done gently in order to dehydrate or cyclize the maleamic acid. The chemical reaction of the amino amino-propionitrile with maleic anhydride to give maleamic acid can be done at room temperature. The organic solvents preferably used are either ether or chloroform. This beta-amino-propionitriles are scarcely found in the commercial sector and their preparation is achieved by the reaction between cyano-ethylation (a by-product of ammonia) and acrylo-nitrile. This reaction is carried out in gaseous phase as well as in liquid phase.
Separation of the product formed is done using the vacuum distillation process Cava, Deana & Mitchell, 1961). Usually, polymers are formed by polymerization or copolymerization techniques. For instance, the N-propionitrile maleimide formed cin the above reaction may be polymerized using the same units or other comonomers added to improve its thermal and chemical properties. Such copolymers have a wide application in the industry especially in the manufacture of electrical appliances due to their ductile nature and thermal stability. Some examples of the monomers which may be used in combination with the N-propionitrile maleimide include the styrene, acrylonitrile, substituted styrenes, maleimide and N-phenyl maleimide.
The polymerization technique employed is similar to that known in the polymerization of maleimide and their derivates particularly the N-phenyl maleimide. In addition, the N-propionitrile maleimides and the N-propionitrile maleamic acid are widely used in the pharmaceutical industries in the manufacture of drugs owing to the nitrile functional group present.
3.1 Maleimides polymerization The research of the novel transparent polymers is an interesting field handled by many investigators. The most common studied polymers are polystyrene (PS) and polymethylmethacrylate (PPMA). Most of these polymers are characterized by lower glass transition temperature and becomes impossible to use them on temperatures above 1000C. Further, great desire has shifted in the heat resistant property of widely used resins such as poly(vinylchrloride) (PVC), polyacrylonitrile-styrene (AS) and polyacrylonitrilebutadiene-styrene (ABS), PS and PPMA. A variety of researches have been carried out to elevate the heat distortion of these resins.
There are two methods commonly used. One involves, blending the resin with a high glass transition temperatures and secondly copolymerization with monomers which has a rigid molecular structure. The first method is economically convenient for small scale application to develop heat resistant reins with good performance balance. On the other hand, the second method is suitable for industrial mass production of the widely used resistant. The monomers commonly used in the second method are α-methylstyrene (AMS) and Maleic anhydride (MAN). These two monomers are employed for the heat resistant in PS and heat resistant PPMA.
Consequently, there are a number of problems associated in the use of these monomers. During the production, large amount of AMS must be incorporated into the polymer structure to enhance the heat distortion of the resins. The incorporation of AMS is difficult because of it has low polymerazability. Accordingly, the heat distortion of the resins is limited due to the fact the amount of AMS incorporated into the copolymer is limited.
Further research has indicated that copolymers containing the Maleic anhydride units tend to decompose during the moulding at high temperature. This is because MAN possesses a low stability in high temperature. Therefore the heat resistant of a variety of resins produced from MAN can be raised to a limited extent as well as the copolymers made from AMS. Due to these problems research has now focused on the use of maleimides. Maleimides are heterocyclic compounds which have a polymerizable double bond and a stable function (rigid structure).
Maleimides are flexible derivatives that find extensive use in biological, pharmacological applications and in chemical synthesis. Maleimides react readily with sulfhydryl groups to form functional stable thioether bonds. This reaction where both sulfhydryl and amine groups are present widely used with proteins. Various results have indicated that maleimides are better monomers than α-methylstyrene and Maleic anhydride when incorporated in the polymer structure. The superior performance of maleimides has been established although not commonly used due to the expensive industrial production of maleimide. A new copolymer developed recently is the fluorinated maleimide that was prepared from fluorinated diol with monofluoronitrobenzene. Another example of copolymer synthesis involves commercial vinyl ether and this maleimide. Most of the copolymers prepared exhibits a glass transition equal to 1650C. One commonly used copolymer is shown below;
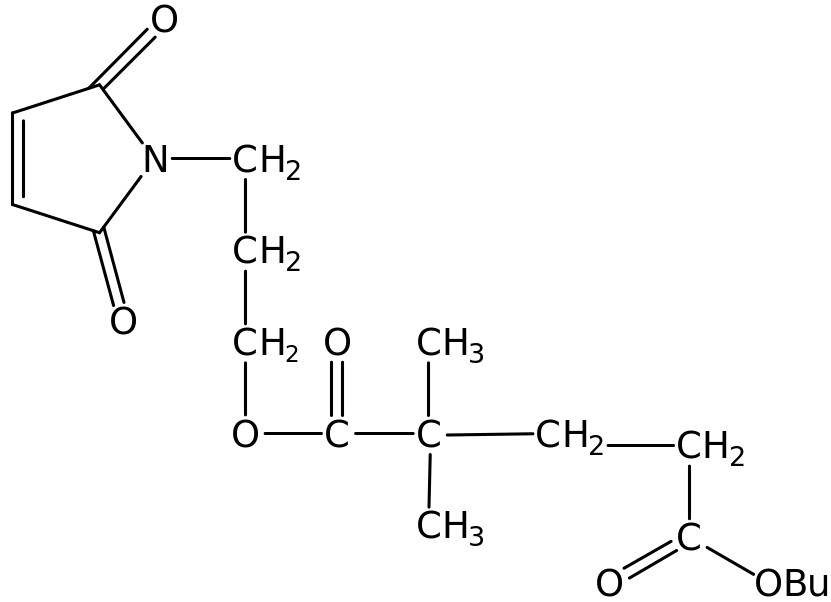
In other technological applications, mono and bismaleimides are widely used in high temperature applications of above 2500C. The rubber is reinforced by maleimides that are used as flexible linking molecules. The double bond readily reacts with amine, thiol or hydroxyl groups to form a stable carbon-nitrogen, carbon-carbon and carbon-sulphur. 3.2 Polymerization of aliphatic maleimides These processes involves the substitution of the H-atom in the maleimide with a straight chain with or without branching or aliphatic chain. The substituent group should posses a reactive molecule (radical) to attach to the NH group of the maleimide.
Mostly the aliphatic chain is either linear or branched containg groups such as methyl or ethyl groups. The maleimide polymers come in different varieties. The mono functional is prepared using one maleimide molecule. When two or three maleimide uints are used, di and tri maleimides polymers result. In other examples, a group comprising of two or more aliphatic maleimide chain may occur. They are connected by spacer groups such as alkyl or cycloalkyl at specific carbon atoms. Spacer group may also consits of oxygen atom readily derived from ethylene glycol and carbonate compounds. The aliphatic maleimide acts as a comonomer as well as a photoinitiator.
The reaction contains photopolymerizable compositions which refers to the compositions which is harden on exposure to sunlight. The reactants include ethylenically unsaturated compounds such as oligomers and monomers which are derived from acyclic and methacrylic acid dissolved in a suitable organic solvent that is copolymerizable with. The reactants must contain the photopolymerizable compound which initiates the reaction when the ultraviolet radiation is passed over the reacting molecules. The photopolymerizable compounds can be polymerized to form homo-polymers or copolymerized with a variety of other monomers.
In photopolymerizable technology, maleimides compounds are used alone or together with other monomers to initiate the reaction of polymerization. The amount of the maleimide compounds required is dependant on the molecular mass of the photopolymerizable being used in the process. The maleimides are known for their reactive terminal, the NH group and are effectively used in coplolymerization to photoinitiate the process.
During the photopolymerizable process, inhibition may occur due to the presence of conventional agents. These inhibitory agents include; UV absorbers, organic peroxides fillers etc. to deactivate the inhibitory processes co-iniators are usually added, for example alcohols, amines and they act as hydrogen donors or electron donors. The co-initiator added should be dependent of the weight of the whole composition of the product. The following structures show a few examples of aliphatic maleimide;
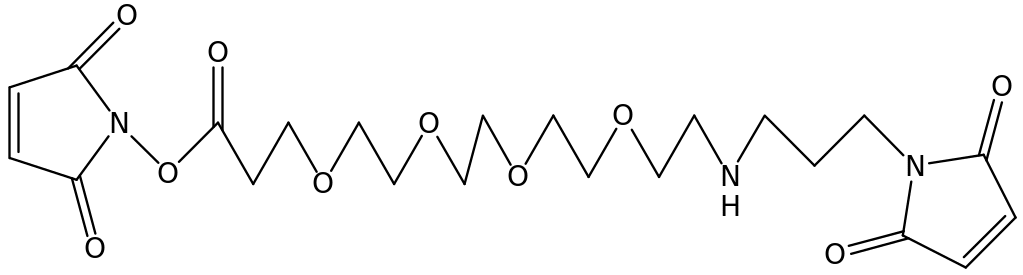
Other examples of aliphatic maleimide polymers have 8 – 12 oxygen atoms.
Maleimide-terminated water-soluble polymers
The attachment of polyethylene glycol to a surface is accomplished through an activated PEG derivative. Such PEG has an activated end suitable for reaction with a nucleophilic centre of a biomolecule such as proteins, cysteine among others. The most common methods used are the reaction between an activated PEG with amino groups such as the abundant Lysine. Despite that many protein molecules retain their activity during the PEG modification, in some situations; the polymer attachment can have undesirable results. Proteins consist of a wide variety of amino acids hence the reactive sites are more than one.
This however, leads to the production of a mixture of polymers consisting of mono-, di-, tetra- etc., which are referred to as pegyglated polymers. This on the other hand becomes difficult to characterization (Gaina & Gaina, 2004, 237). This will consequently affect the subsequent commercialization and the quality of the polymers. This problem can be overcome by the use of a reagent that is selective to a specific site.
The most preferred target is cysteine due to its diol group. Cysteine is less abundant in nature than lysine, thus reducing the likelihood of solving this problem. Polyethylene glycol derivatives are employed in most cases since they contain the diol reactive terminus. Maleimides derivatives such as Polyethylene glycol are commonly used as they contain the site-selective functional group –the sulfhydryl. It is commercially available although the PEG-maleimides is hydrolytically unstable during storage. This instability may lead to the formation of multiple drug-coupling to numerous proteins. Thus there is need of more research to develop or modify the PEG-maleimides with enhanced/ improved stabilities.
References
Cava, M., Deana, A. & Mitchell, J. 1961, N-phenymaleimide, John Wiley & Sons, New York.
Chunqing, L., Huaming, L. & Pengsheng, L. 2004, ‘Free Radical Copolymerization of N-(4-Carboxyphenyl) maleimide with Hydropropyl Methacrylate’, Journal of Macromolecular Science Part A: Pure and Applied Chemistry. Vol. 41, no. 10, pp. 1161-1172.
Gaina, C. & Gaina, V. 2004, ‘Synthesis and Polymerization of Bi-functional Monomers with Maleimide and Allyl Groups’, Designed Monomer and Polymers Vol. 7. No. 3, pp. 235-251.
Hiran, B., Chaudhary, J., Paliwal, S. & Meena, S. 2007, ‘Preparation Polymerization and Characterization of Some New Maleimides’, Journal of Indian Chemical Society, Vol. 84, pp. 385-388.
Oishi, T., Kamori, A. & Fujimoto, M. 1992, ‘Menthylcarboxylatomethyl) Maleimide’, Journal of Macromolecular Science-Pure And Applied Chemistry, Vol. 29, no. 3, pp. 231-247.
Oishi, T. & Fujimoto, M. 1992, ‘Synthesis and Chiroptical Properties of Poly[N-(4-N’-(Alpha Methylbenzyl)Aminocarbonylphenyl)Maleimide]’, Journal of Macromolecular Science-Pure And Applied Chemistry, Vol. A29, no. 12. pp. 1187-1205.
Oishi, T., Kagawa, K. & Fujimoto, M. 1993, ‘Synthesis and Polymerization of N-[[[N’-(Alpha-Methylbenzyl)Amino]Carbonyl]Methyl]Maleimide’, Macromolecules, Vol. 26, no.1, pp. 24-29.
Sharma, N. & Husain, S. 1997, ‘Synthetic Studies of Some Polymaleimides and Copolymaleimides’, Journal of Indian Chemical Society, Vol. 74, pp. 781-783.
Simionescu, I. & Grigoros, M. 1990, ‘Synthesis Of N-Arylmaleimides And N-Aryl-2,3-Dimethylmaleimides’, Journal of Polymer Science, Polymer Letter, vol. 28, no. 6, pp. 39–45.
Wang, H., Wei, J., Jiang, X. & Yin, J. 2006, ‘Novel Polymerizable N-aromatic Maleimides as Free Radical Initiators for Photo Polymerization’, Polymer International, Vol. 55, no. 8, pp. 930-937.