The central purpose of this experiment is to study the primary salt effect in the oxidation reaction of iodide ions to iodine during sodium thiosulfate titration.
Reference to Procedure
All steps of the procedure of this experiment were performed according to the theoretical guidelines given by Amdur & Hammes (1966), Knudsen & King (1938), and Indelli & Prue (1959). Initially, three working solutions and one solvent had to be prepared using distilled water: 250 mL of 0.1 M potassium iodide, 150 mL of 0.001 M sodium thiosulfate, and 300 mL of 0.01 M potassium persulfate. The solvent substance was a complex and contained 0.001 N hydrogen chloride, and 0.00001 M EDTA: 200 mL of 1 M potassium nitrate solution was obtained with the solvent. A mixture of 1 g of starch in 100 mL of water was prepared as an indicator by gradual heating until homogeneous. A series of ten Erlenmeyer flasks are used to obtain different experimental lines, according to the figure below. The start time of each reaction until the blue color appears is considered as ∆t.
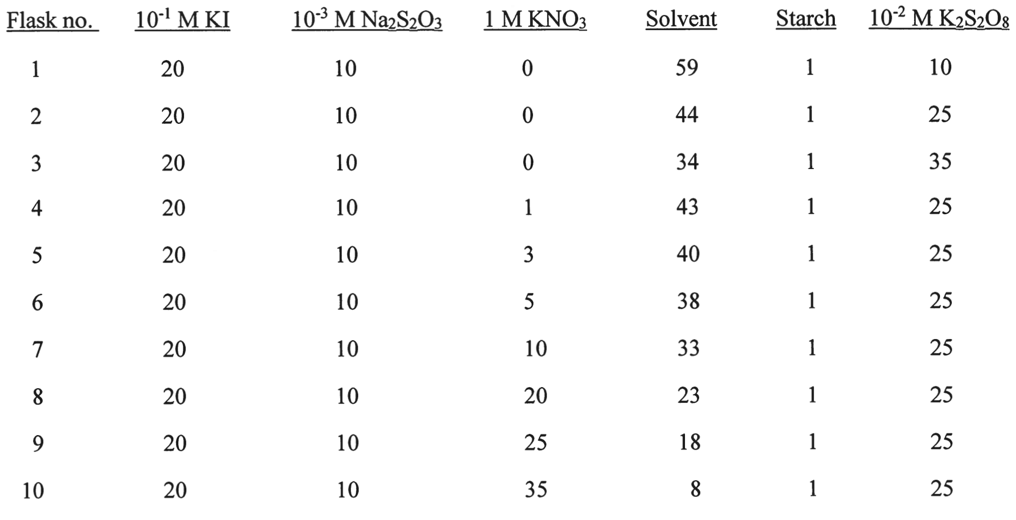
All Chemical Reactions
The Purpose of the Experiment
In ionic systems, there is an effect of increasing the solubility of an ion from a poorly soluble compound when a nonunion of the same charge is introduced into the solution: this is the so-called primary salt effect. Since the iodine clock reaction is used, the reverse reaction for iodide oxidation is the reduction of iodine to iodide with the thiosulfate anion preventing the formation of the triiodide ion. The auto-oscillatory nature of this process is determined by iodine oxidation as a rate-limiting reaction in which the released free iodine is immediately reduced by thiosulfate, and therefore its activity on starch, the appearance of the blue coloring of the solution, cannot be observed as long as the thiosulfate ion remains in the system. In order to speed up the process, a pseudo-first-order method of reaction is used in which the iodide concentration is much higher than the persulfate concentration:
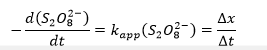
where kapp refers to the apparent rate constant of persulfate, ∆x characterizes the number of added moles of thiosulfate, and ∆t shows the time elapsed from the beginning of the experiment to the appearance of blue staining.
Manipulating the concentration of the persulfate ion leads to the calculation of the reaction rate constant using the Brønsted law. More specifically, the reaction rate, according to Brønsted, should increase when a charge of the same name is introduced into the system and adhere to a linear nature. The change in the reaction rate — the decrease in the time for the blue color to appear — may be due to an increase in the solubility of the ions with the salt effect.
It is well known that the solubility value of low-soluble salts is influenced not only by the electrolytes that share ions with them. On the contrary, the solubility of some low-soluble salts increases in the presence of other soluble salts that do not have common ions with them. Thus, increasing the concentration of potassium nitrate in the experimental series should have reduced the time until the blue coloration of the starch-iodine complex appears. Thus, the essential purpose of the present experiment is to try to evaluate the applicability of the primary salt effect to the iodine oxidation and reduction system, iodine clock, in the presence of negatively charged nitrate ions.
References
Amdur, I., & Hammes, G. G. (1966). Chemical kinetics. McGraw-Hill Books Co.
Indelli, A., & Prue, J. E. (1959). 23. Kinetic salt effects in the reaction between persulphate and iodide ions. Journal of the Chemical Society (Resumed), 107-111.
Knudsen, O. M., & King, C. V. (1938). The oxidation of iodide ion by persulfate ion. V. The rate at low ionic strength. Journal of the American Chemical Society, 60(3), 687-691.