Introduction
The management of resources, which are freshly extracted from the nature and those that are discarded influence the present environmental conditions and the future availability of natural resources largely (Kollikkathara, Feng, & Stern, 2009). However, the traditional importance that has been given on the exploitation of natural resources to manufacture products that are ultimately discarded as waste after their useful life has resulted in dramatic increase in waste throughout the world (Kollikkathara, 2009) (Kollikkathara et al., 2009). A consequent observation is the fact that waste generation and management have multiple consequences with respect to socio-economics, human health, aesthetics and amenities for people (Louis, 2004)
At the same time, the demand for energy has been dramatically increasing over the years, compelling nations to adopt newer and effective energy conservation and generation technologies to cope with the situation within the time of a generation (Porteous, 1998). Consequently, increasing focus has been given in recent years on the generation of energy from waste. To be precise, authorities such as the SWA (Solid Waste Authority) of the US are focusing on integrated waste management (IWM) including waste-to-energy activities along with reuse, recycling and recovery practices (Kollikkathara, et al, 2009). In order to understand the concept of waste-to-energy, it is essential to focus on the sources of waste, and the importance and methods of energy generation from waste.
Waste to Energy – an Overview
Waste to energy process involves the conversion of various elements of solid wastes like paper, plastics and woods for generating energy. This can be accomplished by either a thermo chemical or a biochemical process. Before considering any application of the waste to energy technologies, it is imperative that a comprehensive waste management strategy is developed to consider the cost-benefit of the proposed project. The economic feasibility of any waste to energy project depends on the total volume of waste generated and the cost of managing the waste. The waste management cycle normally involves different stages like collection of waste, transportation and disposal of the waste material collected. The disposal method is one of the important step in the waste management programs as it affects the environment and costs of managing the waste. The cost of transporting the waste is another important element that needs consideration in the process of converting waste into energy.
There are three important ways of disposing the waste recognized. They are:
- use the waste for landfilling purposes,
- converting waste into usable material through a process of recycling
- converting waste into energy.
Generally, landfilling option is considered the most economical way of waste disposal. If the tipping costs are low and the disposal, sites are within a close range this option can be considered in preference to other ways of disposing the waste. Similarly, the market for recycled waste materials needs to be assessed for considering the option of recycling the waste to produce new usable materials. Presently since there is no viable market for any recycled waste, material except for aluminum, there seems no point in establishing a recycling program with a significant cost involved. It is to be considered whether the recycling programs generate revenue at least to meet the cost of collecting the waste. Therefore, a waste to energy plant is a viable alternative to develop a solid waste disposal plant. A waste to energy plant would be able to reduce the volume of waste by almost 90 percent. If the costs in disposing the refuse increase to a point when it is no more effective to adopt the landfilling method, it is advisable to consider a waste to energy application. Considerable disposal cost can be saved by adopting a waste to energy conversion method for waste disposal. The objective of this paper is to examine the different methods of converting the waste to energy and the relative merits and demerits.
Statement of Problem
Industries in both the economically advanced and developing countries generate large quantities of solid and liquid wastes. In addition, people living in larger cities generate significant volumes of solid and liquid wastes. Most of these wastes generated are not treated properly and they enter into land and water bodies. Since the wastes are not treated, they tend to emit gases like Methane (CH4) and Carbon Dioxide (CO2). These gases in turn produce odor and cause air pollution issues. Problems and issues created by wastes can be mitigated by adopting environment-friendly waste-to-energy technologies. These technologies help in proper treatment and processing of the wastes before they are disposed of. Such treatments reduce the volume of waste that need to be disposed off and help in controlling the pollution issues. There is a substantial quantity of energy also produced during the process of treating the wastes, which adds to the economic growth of the nation concerned.
There are a number of constraints and barriers to the adoption of processes of waste to energy. First, there is a clear lack of cohesive leadership at the Federal, state and regional levels to promote the technologies for converting waste to energy. In addition, it is to be acknowledged, that there are no statutory frameworks for promoting conversion technologies. Similarly, there are no statutory disincentives for not adopting any of these technologies. Limitations on funding and the absence of proven technologies, which demonstrate the efficacy of such technologies, is another barrier to the adoption of the process for converting waste to energy.
In some cases, expensive development costs and large-scale sizes make these technologies too risky and make their adoption unviable. Moreover, under current marketing conditions, the conversion technologies have been found to be uncompetitive and the costs of renewable energy from the waste deters the public from making use of the renewable energy Lack of reliable and complete data on the lifecycle benefits act as deterrent for the prospective use of the conversion technologies. Unavailability of information on the performance of waste to energy technologies and unavailability of feedstock and lack of details on vendors also prohibit the conversion of wastes to energy.
Within this context, the objective of this study is to report on the various technologies available for conversion of waste into energy, the costs and benefits of these technologies and the problems and issues associated with the adoption of these technologies. The economic impact of the generation of wastes and the adverse effect of wastes on the environment is also presented.
Hypothesis
The awareness and attitude towards investments and benefits of renewable energy generated from waste is different in the people among different income groups and people living in rural and urban areas.
The thesis will focus on proving the null hypothesis that the awareness on the benefits of converting waste into energy is different among the people belonging to different income groups. The assumption here is that the knowledge and attitude of the people in the lower income groups is better since the rising energy prices hit them more strongly than those belonging to higher income groups. Similar is the case with the people living in rural areas where the knowledge to put the waste into alternative uses is more than the people living in the urban areas are.
Significance of the Study
The quantum of waste produced in both industrially advanced and developing countries are enormous and the disposal of the waste created becomes a costly affair. Moreover, large expanses of otherwise usable land areas are being consumed for disposing the wastes. This not only causes economic loss but also creates high pollution. Especially toxic wastes like glass, processed chemicals produced by industries, plastics, foams and poisonous gases are perpetually polluting the earth leading to imbalances in the ecological system. Therefore, there should be some effective ways of managing the waste so that the cost involved and the volume of waste is reduced to the minimum, Conversion of waste to energy is identified to be one of the effective ways of waste management. The conversion of waste to energy produces significant benefits to the society including prevention of the release of greenhouse gases, providing an alternative form of energy, reducing the impact on landfiils used for refuse disposal and saving the natural resources of earth by using less oil, coal, or natural gas for electricity generation. In view of these tangible benefits to the society, this study on conversion of waste to energy assumes significance. This study examines the waste to energy processes to enlarge the existing knowledge in the field.
Aims and Objectives
The aim of the research is to examine the process of conversion of waste to energy with specific reference to the benefits resulting there from and the barriers to adopt an effective waste to energy technology. In the process of achieving this aim, the study covers the following objectives.
- To study the various technologies available for conversion of waste into energy, the costs and benefits of these technologies and the problems and issues associated with the adoption of these technologies.
- To elucidate the economic impact of the generation of wastes and the adverse effect of wastes on the environment
- To examine the efforts in the direction of conversion of waste to energy in different countries
- To recommend a few possible ways of effective waste management, based on the knowledge acquired out of the review of the related literature
In order to present a comprehensive report, this thesis is structured to have different chapters. The first chapter in addition to introducing the subject of conversion of waste to energy also presents the aims and objectives of the study along with the hypothesis to be proved/disproved. Chapter two makes a detailed review of the related literature to provide an in-depth knowledge on the subject of study. The methodology used for conducting the study is described briefly in chapter three. While chapter four contains the findings of the research followed by a detailed analysis of the findings, chapter five makes the concluding remarks with few recommendations on effective waste management.
Review of the Related Literature
The objective of this chapter is to provide an extended knowledge on the topic of conversion of waste to energy through a review of the available literature. This chapter will cover the economic impact of waste management, technologies available for the conversion of waste to energy, merits and demerits of the different technologies and the barriers to the adoption of these technologies. The review would also bring out the waste management practices in different countries.
Introduction
In almost all the countries of the world, millions of tons of solid and liquid waste materials are produced by households as well as industries. Although these wastes materials have a significant energy content, they are being disposed in landfills. Growing landfill charges and other ways of managing wastes increase interest in the recycling and using waste as a potential source of energy. The basic purpose of waste management principles aim at preventing the generation of waste, to make use of the waste as recycled usable material and using waste as a source of energy. The most important objective of waste management is to carry out safe and effective methods of disposing the unusable waste. “The most important aim over the next few years will be to develop source-separation methods for waste in order to direct waste both for material recovery and for energy use.” It is possible to use a sizeable volume of the waste for generating energy for generating energy, but this involves the development of different technologies for new waste flows and power plant systems. Since the costs of separating the waste can cover the total quantity of waste materials, material recycling can also be focused along with the increased use of waste for energy creation. The following picture illustrates the waste to energy problems and benefits.
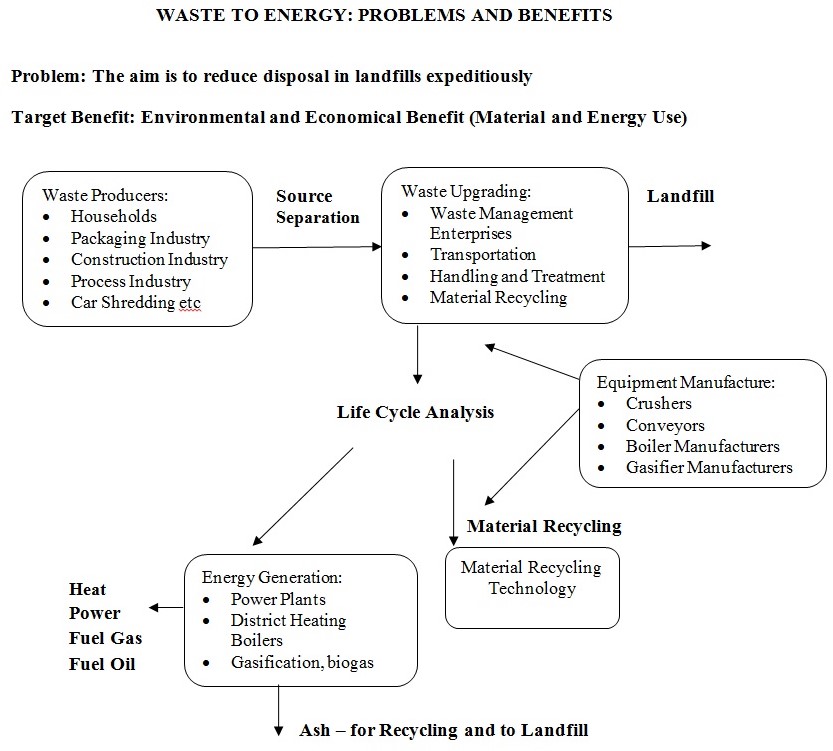
This chapter presents a detailed outline of the issues involved in the conversion of waste to energy.
Sources of Waste
There are various sources for waste generation, from industry and agricultural to household activities of humans (Sources of Waste, 2002). More precisely, activities pertaining to agriculture, forestry, extractive industry, urban development and homes result in huge amounts of solid waste, sediments along with nutrients and toxic pollutants. A discussion on the different sources of wastes is produced below:
Extractive Activities
Extractive and refining activities related to materials such as oil and gas result in the release of large amounts of waste, especially into the marine environment due to the use of oceans as dumping areas or accidental spillage of toxic materials (Sources of Waste, 2002).
To be precise, research on a copper-mine in Vancouver Island of Canada revealed the loss of biodiversity in the region due to the release of nearly 50,000 tons of silt per day for 25 years (Sources of Waste, 2002). However, the biodiversity was regained slowly after the deposition of silt was ceased (Sources of Waste, 2002).
Household Activities
These activities, in the urban areas particularly, result in the release of different materials such as sewage, solid waste, toxic pollutants and greenhouse gases (Sources of Waste, 2002). According to the Department of the Environment, Food and Rural Affairs in the UK, nearly 28.2 million tons of municipal solid waste (MSW) was produced domestically during 2000-2001, of which 89 percent came from households (Barr, Gilg, & Ford, 2005). The MSW had increased at an average rate of 3% from 1996 to 2001, with 78% of total MSW being sent to landfill and only 12 % being recycled (Barr et al., 2005).
Manufacturing Activities
The manufacturing industry includes a broad range of areas, the activities of which lead to enormous amounts of solid waste (Sources of Waste, 2002). For instance, the chemical industry encompasses the productions of over 100,000 chemicals, with over one thousand tons per year production of nearly 1000 chemicals, related to categories such as agriculture, paper and pulp industry, textiles, detergents and cleansers, plastics, paints, pharmaceuticals, solvents, propellants, and refrigerants (Sources of Waste, 2002). Furthermore, the cement industry, which is vital for the urbanization and industrialization processes, is one of the largest producers of CO2, contributing up to 3% of all CO2 emissions (Sources of Waste, 2002).
Rural Land Use Activities
Agriculture is the major source for waste generation in rural areas (Sources of Waste, 2002). Many of the waste materials resulting from the use of pesticides, herbicides and fertilizers are toxic pollutants such as aldrin, chlordane, DDT, dieldrin, endrin and hexachlorobenzene (Sources of Waste, 2002). Moreover, the increasing restrictions on the use of such toxins are unable to maintain pace with the development of new compounds (Sources of Waste, 2002).
Also, the conversion of forests into natural lands has led to severe losses of standard biomass, contributing to global climate change (Sources of Waste, 2002).
Transport
By the end of the twentieth century, the increase in the number of vehicles all over the world reached a rate of nearly 3% per year, approximately double the rate of population growth (Sources of Waste, 2002). Apart from the problem of increasing air pollution from these vehicles, the relatively short life span of many of these vehicles is leading to dramatic increase in the amounts of solid waste as well by the shredding of the old and unusable vehicles (Sources of Waste, 2002).
Municipal Solid Waste (MSW)
It is a well-known fact that urbanization and industrialization have resulted in enormous increase in the amounts of waste generated (Kollikkathara et al., 2009). Accordingly, proper management of municipal waste collected from densely populated urban areas has become a major problem in developed countries due to high awareness pertaining to environmental sustainability, and material and energy recovery (Kollikkathara et al., 2009).
MSW consists of dust and cinder, vegetable matters, paper and cardboard, textiles, glass, plastics and other materials, with paper and cardboard constituting majority of the waste materials. Analysis of the chemical composition of the MSW shows that the waste contains gases like hydrogen, oxygen, nitrogen, chemical substances like carbon, sulphur, and chlorine, water, combustible materials, ash and inert and moisture among several other materials with carbon, water and ash and other inert materials constituting the majority of the substances (Porteous, 1998).
Depending on the predominant material the composition and calorific values of MSW may vary. For instance, the calorific value of paper and plastics-rich wastes can be as high as 14 to 17 MJ/kg (Porteous, 1998, p. 180). Also, most of the carbon content of MSW (85%) is derived from paper and vegetable matter, and its combustion to CO2 is CO2 neutral, making CO2 available to plant growth (Porteous, 1998). Moreover, Porteous (1998) states that the waste-to-energy process results in increased CO2 reduction benefits, as the combustion of MSW for this purpose prevents the formation a potent greenhouse gas, methane, which was generated from landfills at a rate of 2.5 mt/yr as per 1990 estimates (as cited in Porteous, 1998, p. 181).
Wastewater
It is a combination of sewage, storm water and water, which is used for various purposes in residential and non-residential sources (Wastewater, 1997). Domestic sewage can be classified in to black water or wastewater from toilets, and gray water, which is the wastewater from sources other than toilets (Wastewater, 1997). Non-residential sources of wastewater vary among a vast range of areas from offices to public rest rooms to industries (Wastewater, 1997). Accordingly, the quantity and composition vary depending on the source (Wastewater, 1997). To be precise, wastewater from restaurants mostly contains oil and grease, whereas different manufacturing industries may release various chemical and biological pollutants in wastewater (Wastewater, 1997). Improper of treatment of wastewater can harm public health as well as the environment (Wastewater, 1997).
Though the constituents of wastewater vary depending on the source of generation, they normally encompass organisms including pathogens; organic materials arising from human wastes, paper products, detergents, cosmetics, foods, and from agricultural, commercial, and industrial sources; oil and grease; inorganic minerals, metals and compounds; nutrients such as nitrogen and phosphorus, solids; and gases (Wastewater, 1997).
Certain tests are used to evaluate the strength of wastewater, and the intensity of treatment required, periodically measure the quality of effluent and quality of receiving waters (Wastewater, 1997).
- Biochemical Oxygen Demand (BOD) – It is used to measure the strength of wastewater with respect to the amount of dissolved oxygen that is required by organisms to degrade wastes (Wastewater, 1997).
- Total Suspended Solids (TSS) – It is useful in indicating the quality of wastewater and its impact on the environment, while clarifying how much secondary treatment is required to treat the wastewater (Wastewater, 1997).
- Total Coliforms and Fecal Coliforms – It is helpful in observing the effectiveness of treatment processes (Wastewater, 1997).
Treatment of wastewater results in the production of huge amounts of sewage sludge, which can be used as a biomass resource for the production of energy (Rulkens, 2008; Boran, Houdkova, Uckaj, & stehlik, 2008). The method of energy generation from wastewater will be discussed in subsequent sections.
Sustainable Waste Management
The first initiative in the direction of sustainable waste management was taken at the United Nations Conference in Stockholm in the year 1972. Subsequently a majority of the governments introduced measures for sustainable waste management. Sustainability, self-sufficiency and the proximity principle and the waste hierarchy are the key principles that underpin the waste strategies of major governments. The waste hierarchy depicted in the picture below summarizes the key objectives of sustainable waste management. The objectives are:
- To minimize the amount of wastes generated
- To optimize the amount of material that can be reused or recycled and
- To minimize the amount of waste disposed in landfill
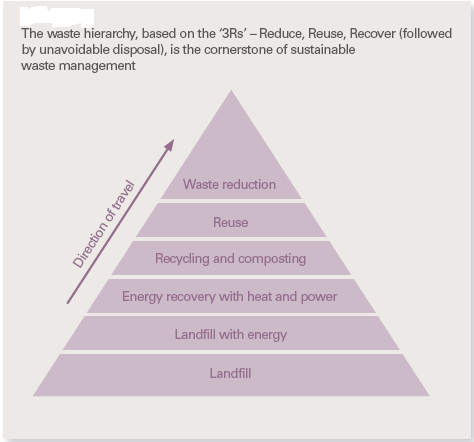
The aim of sustainable waste management is to move from the bottom level of the hierarchy and to eliminate landfill as much as possible.
Urbanization and Waste Generation
Urbanization contributes to generation of waste and unscientific ways of handling waste would cause health hazards and urban environment degradation. “Municipal Solid Waste is defined to include refuse from the households, non-hazardous solid waste discarded by the industrial, commercial and institutional establishments, market waste, yard waste and street sweepings which are collected by the municipal authorities for disposal”. In any advanced economy, MSW represents only a minor portion of the total waste generated in the country. Post-Consumer waste is the main focus for Municipal Solid Waste Management with measures for prevention, treatment, recycle, reuse and disposal.
Impact of Unscientific Handling of Waste
Unscientific handling of MSW and other industrial waste lead to adverse health and environmental impacts. Indiscriminate dumping of the solid waste leads to several consequences like:
contamination of ground water by the leachates generated by solid waste dumped unscientifically,
- contamination of surface water by the runoff from the waste dumps,
- generation of foul odor, pests, rodents and windblown litter in and around the waste dumped,
- generation of inflammable gas (methane) within the waste dumps resulting in fire, smoke and smog in and around the landfill where the solid waste is dumped,
- releasing of greenhouse gases like carbon-dioxide and methane,
- generation of epidemics through stray animals and other bacteria causing various diseases,
- in large settlements and colonies of the developing countries, inefficient waste management has been the prime reason for serious urban pollution and health hazard (Jain, 2007).
Recovery
Recovery of materials can be carried out through various processes such as recycling, composting and certain energy recovery processes.
- Recycling: This process involves considerable environmental consequences and costs with respect to the materials and methods chosen for the process. In addition, it suffers from challenges such as market availability, recyclability of products, market prices, collection, processing and transportation costs, and the lack of public participation and education.
- Composting: It also has certain problems on the part of quality and bulk marketing, apart from other challenges described above. However, involvement of farmers can help in the growth of this approach.
- Energy from Waste: Energy recovery should be given importance along with recycling and composting, failing of which may lead to the loss of nearly 500 kWh of electricity per ton of waste. In addition, the possibility of bottom ash recycling helps in reduction of ash residues for disposal into landfill to less than 10 percent (Porteous, 1998, p. 178).
A model diagram depicting waste to energy technology is shown as follows. The details of various processes for the recovery of different forms of energy will be discussed further.
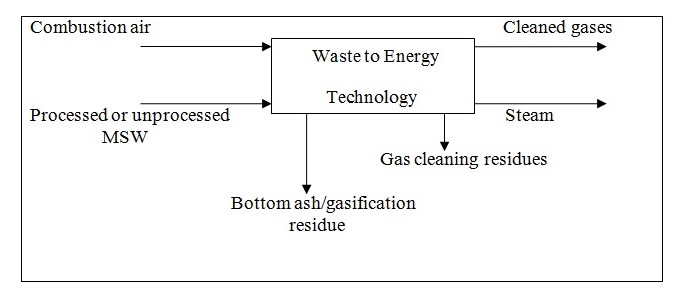
With an objective to focus on the need for effective energy/heat recovery from waste management and utilize it for enhanced power production so as to reduce dependency on fossil fuels, minimize pollution and increase energy efficiency of the WtE plants, Swithenbank et al. (2000) conducted study evaluating various options of integrating waste management with power generation methods. According to the authors, the estimated total heat value of municipal waste in Britain was equivalent to nearly 30% of that of the coal required for energy needs (Swihenbank, Nasserazadh, Wasanakorn, Lee, & Swithenbank, 2000). Importantly, the electrical production efficiency of coal burning plants and waste incinerators were only 37% and 20%, respectively. The authors formulated that an integrative approach such as combining steam from waste incinerators with the steam portion of a gas-fired combined cycle would help achieving more energy efficiency than that attained by respective individual fuel utilization systems. Moreover, the use of a high temperature heat exchanger in a recuperative gas turbine can enhance the generation efficiency to 60% without the need to use a steam cycle. Furthermore, adopting an integrated pyrotechnology system in which the steam from a conventional waste incinerator is gasified with coal to release a combination of carbon monoxide and hydrogen that can be used in a combined gas turbine/steam system (Swithenbank et al., 2000).Waste-to-Heat and Electrical Energy: Feasibility and Efficiency Studies
They stated that unlike the combustion of fossil fuels, combustion of organic materials of the waste would not result in net release of CO2 in to the atmosphere. Furthermore, materials that cannot be recovered such as certain forms of plastics can be burned to recover their heat values. However, the ability of high flue gas temperatures to corrode boiler parts requires maintaining low possible temperatures, thereby resulting in only 20% energy efficiency of waste incineration. They further mentioned that energy from waste incineration that can be used for electricity generation is more valuable than its value as heat. Hence, maximizing the potential for electricity generation from waste incineration requires considering integrative approaches such as co-incineration of waste with fossil fuels. In this regard, two alternatives were proposed to increase the steam temperature, namely coating the metal tubes of the boiler with corrosion resistant materials such as ceramic and using a boiler with conventional fossil fuel to superheat the steam from the incinerator to rise its temperature to nearly 565 0C, thereby making it more efficient for electricity generations. In addition, the large temperature difference between steam from the incinerator and the boiler using fossil fuel minimizes the need for supplementary fuel to maintain required steam temperatures (Swithenbank et al., 2000).
Furthermore, an integrated system of steam maintenance for power generation uses intercooler, recuperator and reheat for rising the temperature of steam that is transferred from waste incinerator to a simple combined cycle of power generation containing gas turbines with air cooled blades along with two steam turbines. Optimization of the process stages helps in high overall efficiency for electricity generation. The use of a high temperature heat exchanger was also considered by the authors, as such an exchanger can reduce the space required for a WEP (Waste, Energy and Pollution Management System), by replacing gas turbines, multi-stage turbine reheat and cooling towers with an optimized gas turbines, high temperature recuperator and a single-stage turbine reheat. This system can increase the energy efficiency up to 60%. Moreover, as the heat exchanger works at modest pressure ratio of 5 to 10, NOx emissions will also be reduced during the process (Swithenbank et al., 2000).
The authors further focused on some more alternatives such as co-gasification systems and coal gasification using steam from the incinerator. To be precise, gasification of coal for electricity generation requires water; and sewage sludge contains nearly 70% water and 30% solids. Hence, the sludge can be utilized for coal gasification, thereby transferring the energy of the sludge solids to contribute to the energy of the gas product. In addition, the conversion of steam from waste incineration into gas and its utilization in coal gasification enhances the electricity generation efficiency to 60% in comparison to 20% efficiency of conversion of steam directly into electricity. Co-incineration is another technique that can be used to incinerate tyres. It was estimated that developed countries contained at least one waste tyre per year per adult person, and direct incineration of these tyres would pose problems due to the embedded steel wires. On the other hand, co-incineration of these tyres with a fossil fuel in a cement kiln acts as a safe disposal method while reducing the demand for fossil fuel. The materials of the tyre such as iron, calcium and sulphur can be safely tackled as they are incorporated into the cement clinker (Swithenbank et al., 2000).
Like this, Swithenbank et al. (2000) focused on various means of integrated approaches that could possibly enhance the electricity generating efficiency of waste incineration and conventional power generation methods using fossil fuel.
Generation of Thermal Energy from MSW
Dajnak & Lockwood, (2000) examined the use of thermal energy from municipal solid waste to desalinate seawater. The article was published in the journal of Desalination in 2000. According to the others, many regions throughout the world have a combination of waste disposal and water resource problem. The use of thermal energy from waste incineration for seawater desalination can help reducing the above two problems. To be precise, several leisure communities developing in Egypt away from the Nile face problems of waste disposal and water resource due to their isolation from water available areas. This problem can be alleviated by adopting the stated approach (Dajnak & Lockwood, 2000).
Desalination of water is defined as a process of removal dissolved mineral from seawater, brackish water or treated wastewater. In 1993, more than 15Mm3/d of fresh water were produced worldwide by desalination processes. Depending on the site and economics, desalination can be carried out through distillation, electro-dialysis or membrane techniques. Distillation is carried out by boiling seawater and collecting fresh water by condensing the vapor that is free from minerals. It can be either multiple effect distillation (MED) or multistage flash (MSF). Electrodialysis is the process in which ions forming the salt are removed by electrical forces and concentrated in separate compartments. However, electrodialysis requires more electrical power than other processes with increasing salinity of the raw water. Reverse osmosis (RO), known as hyperfiltration and ultrafiltration, is the process of forcing water molecules through a semi-permeable membrane, as the salt molecules cannot pass through the membrane. As the RO is less heat intensive, it does not have the problem of corrosion that is often seen with distillation, and, unlike remaining methods, RO can be started-up and shut down without much time lag (Dajnak & Lockwood, 2000).
The electrical consumption of RO plants ranges between 3 and 9 kWh/m3 of fresh water produced. As per the author’s estimations, the calorific value of MSW was 8.5-12.8 MJ/kg that could produce 3.4-5.1 MJ/kg of electricity. Accordingly, it was estimated that 188L of fresh water could be produced through desalination of seawater in RO technique with the incineration of only 1kg of waste. Hence the authors concluded that use of thermal energy from waste for seawater desalination, by RO particularly, had merit for consideration as the process has remarkably little air and water environmental impact (Dajnak & Lockwood, 2000).
Process of Pyrolysis
Phan, Ryu, Sharifi, & Swithenbank, (2008) examined the efficiency of pyrolysis of segregated wastes, namely waste wood, cardboard and textile residues, for energy production.. According to the authors, energy from wastes can be recovered by thermal or biological procedures. Thermal processes are useful in case of mixed wastes, whereas biological wastes are preferred for wet organic wastes such as food waste. The authors stated that though the industry mostly prefers incineration method of thermal processing, pyrolysis and gasification methods have higher energy efficiencies and flexible uses of the fuels derived (Phan et al., 2008).
Pyrolysis is the thermal decomposition of high molecular weight compounds of wastes into smaller fragments through dehydration, decarboxylation at low temperatures and the formation of free radicals at temperatures of over 350 0C. Consequently, raw materials are converted into reactive intermediate products such as char and light molecular weight gases, which will be condensed with cooling. The process leads to great reductions in polymer lengths from 10,000 untis to 200 units at temperatures above 300 0 C. An increase in pyrolysis temperature leads to decreased yield of char and increased yield of gas. In addition, the liquid yield attains peak at nearly 500-600 0C, beyond which it decreases because of thermal breakdown of heavy compounds. Moreover, some gasification procedures use pyrolysis as a pre-processing stage, after which the heavy compounds will be broken into gases through thermal and catalytic cracking (Phan et al., 2008).
However, the problem for employing pyrolysis for MSW is the highly inhomogeneous nature of mixed wastes, which contain different types and sizes of materials along with ash, heavy metals and chlorine, thereby increasing the operating cost and reducing the quality. Nevertheless, certain combustible portions of segregated wastes, such as cardboard, waste wood, textiles and plastics, without economic viability for recycling can be processed through pyrolysis (Phan et al., 2008).
The authors used mixed waste materials of cardboard, waste wood and textile residues collected from an urban area to pyrolyse in a fixed bed reactor with a sample weight of around 200 g and test the efficiency of pyrolysis. The equipment included a stainless steel reactor placed in an electrically heated furnace, and was connected to two water-cooled condensers (Phan et al., 2008, p. 66). Each time, 150-200 g of sample was heated from room temperature to final temperatures of 350-700 0C and kept there for two hours for allowing enough time to complete pyrolysis. The gas sample was taken from the condensers to a MDA 3000 CO/CO2/O2 analyzer and for analysis with a Varian CP-3800 GC; whereas the char and liquid products were collected when the reactor cooled down (Phan et al., 2008).
The pyrolysis of three segregated waste materials revealed that char constituted around 38-55% of the energy content of original materials, whereas the liquids had 20-30%. The carbon content and calorific value of char increased with temperature, but the mass and energy yields slowly decreased. Pyrolysis liquids had an average calorific value of 10-12 MJ/kg, and contained large amount of water and many oxygenated compounds such as furans, derivative carbonic acids and anhydrosugars. The aqueous fraction of the liquid stood at nearly 33% of the initial sample, and rise in temperature beyond 600 0C reduced yield due to thermal and catalytic cracking. As a whole, the authors concluded that pyrolysis temperature for producing char and liquid should be less than 500 0C to maximize the energy yield. The pyrolysis gases contained mostly CO and CO2 with increasing yield of methane and hydrogen with the rise in temperature (Phan et al., 2008, p. 70). The calorific value of dry pyrolysis gases was about 13-16 MJ/N m3 at 700 0C (Phan et al., 2008).
Process of Incineration
Previously, Bebar, Martinak, Hajek, stehlik, Hajny, and oral, (2002) worked on the aspect of energy consumption in the process of utilization of the heat value of thermal decomposition products of waste. This work was with respect to two different waste processing technologies-the first being an incineration plant working under oxidizing regime and the second was an incineration plant containing a gasification reactor for the first stage of the process.
According to the authors, heat in thermal decomposition of waste is produced by burning the combustible materials of the waste and the burning of an auxiliary fuel while processing waste with low heat value. They stated that implementing a two-stage incineration for the thermal treatment of waste was required while ensuring the second stage to release flue gas temperature as per related regulations. Also, it is essential to maintain a minimum residence time of the decomposition materials in the second stage/after-burner chamber. Different countries set various recommendations in this regard. To be precise, the EU directive requires maintaining the after-burner temperatures of 850 and 1100 0C depending on the amount of chlorine in the waste at less than or more than 1 wt% (Bebar et al., 2002, p. 898). Also, 6 vol% of surplus oxygen should be maintained in the flue gas from the after-burner chamber so as to enable proper combustion. The authors also stated that the first phase of combustion can be performed in the oxidizing or a pyrolysis regime, the only difference between the two methods being in the requirements of heating-up the products of the first thermal decomposition phase to the temperature required in the after-burner chamber (Bebar et al., 2002).
They compared the process and related costs of the incineration with oxidizing and gasification methods. For this purpose, they assumed incineration plants with an annual waste capacity of 10 kton while operating for 7000 hours per year. The heat values of waste for processing were considered as 30 and 15 MJ/kg. For the first method, they examined two thermal regimes, one with 8700C flue gas temperature at the rotary kiln exit and the other with the related temperature equal to 10500C. They estimated the consumption of auxiliary fuel by observing the temperature of the flue gas released from the after-burning stage. For examining the second method, incineration plant containing gasification reactor, they considered two types of waste same as above. While maintaining stable flue gas exit temperature of 850 0C and supply of 35% of the stoichiometric quantity of air, additional steam of 0.4 kg/kg was supplied for gasification of the more energetic waste. They assumed that the total steam would be used for power generation, thereafter for technological heat-up. The net profit of power generation was estimated by subtracting the consumption of the incineration plant from the total power generated (Bebar et al., 2002).
The results of the experiment revealed that the use of oxidation technology for the thermal treatment of wastes results in large production of flue gases and heavy consumption of auxiliary fuel in the after-burner chamber. This consumption of auxiliary fuel can be minimized by employing gasification technology in the first phase of the treatment process. For the gasification technology allows thermal fission of the waste at temperatures below the melting point of ash, hence decreasing the consumption of auxiliary fuel by using the heat value of fission products from the gasification reactor (Bebar et al., 2002, p. 906).
Energy Efficiency of Material Recovery to Waste Incineration
A study to compare the energy efficiency of material recovery to waste incineration with energy recovery was carried out by Holmgren & Henning, (2004) by observing two Swedish municipalities. The authors pointed out that material recovery can save virgin materials and energy, as manufacturing processes using recovered materials are less intensive than procedures using virgin materials. The type of process for more energy efficiency also depends on the kind of waste material. For instance, metals and glass save energy when used as recycled material than incinerated, as they do not make any heat contribution; whereas materials such as cardboard, paper, plastic and certain biodegradable waste can be processed effectively in both approaches (Holmgren & Henning, 2004).
According to the authors, the hierarchy of waste treatment practices as stipulated by the European Commission is waste prevention, recovery and disposal, while giving preference to material over energy recovery. In addition, the Swedish Ordinance on Producer Responsibility encourages material recovery over energy recovery. This tendency was further evidenced by the governments study to evaluate the feasibility of imposing taxes at various stages of incineration to promote material recovery than energy recovery for achieving improved energy efficiency. However, both material and energy recovery methods have advantages in terms of savings of virgin materials and energy, and replacement of primary energy sources with renewable sources. Hence, the authors examined the energy efficiency by optimizing and observing the recovery processes in two municipalities with respect to the amount of waste incinerated and fuel replaced by waste, and comparing it to possible benefits of material recovery from the same waste (Holmgren & Henning, 2004).
The municipalities studied were Linkoping and Skovde. Linkoping had an existing waste incineration plant with a capacity of 250000 tons of waste per year and was about to increase its capacity to 350000 tons; whereas Skovde was planning to establish a waste incineration plant with a capacity to process 52000 tons of waste per year. The purpose of these plants was to supply energy to respective district heating systems. While comparing material recovery to energy recovery, the authors also focused on the environmental effects with respect to carbon dioxide emissions by observing the type of fuel that the waste replaced and the relative amount of electricity produced (Holmgren & Henning, 2004).
They used an optimization model, called MODEST, for optimizing dynamic energy systems flexible time divisions and boundary conditions to give consideration for timely variations in energy requirements and material supply in waste. They focused on two scenarios, material recovery without energy recovery and energy recovery, in the two municipalities to evaluate respective financial prospects and energy efficiency. They adopted various assumptions with respect to suitable treatment of different material fractions in each scenario. To be precise, paper, cardboard, hard plastics, glass and metals were assumed to be material recovered in material recovery scenario, whereas all waste was assumed to be incinerated in the energy recovery scenario. They also considered factors such as the amount of recycled material needed in excess to get the strength of a cardboard similar to that made by primary material (Holmgren & Henning, 2004).
The observations in both scenarios for various material fractions revealed interesting facts with respect to the use of suitable energy efficient methods for various material fractions. It was observed that the application of material recovery and/or energy recovery options largely depends on the type of material fraction. For non-combustible materials, material recovery should be adopted, as their combustion does not yield any net gain in energy; instead material recovery saves both virgin materials and energy. To be precise, incineration of iron and glass results only in bottom ash without any energy production. In contrary, recovery of metals can save both material resources and energy required to process virgin materials/ores. Furthermore, though energy savings from recovery of some materials such as glass are comparatively small, they should be opted out from incineration in order to conserve space for accommodating combustible materials which can yield energy (Holmgren & Henning, 2004).
In contrary, certain combustible materials pose a problem in deciding the better waste management, as they can be either material recovered or energy recovered. In such instances, potential energy savings by both processes need to be compared. For instance, material recovery from cardboard is less efficient than energy recovery, as the production of cardboard from recycled material requires more electricity and material than from virgin material. On the other hand, material recovery of paper is advantageous than energy recovery from it, as paper production from recycled material takes less electricity and nearly equal amount of material compared to production from virgin material. Furthermore, biomass should be saved to the extent possible, as it replaces fossil fuel combustion (Holmgren & Henning, 2004).
Environmental Impact of Waste-wood Co-Utilization
Skodras, Grammelis, Kakaras, & Sakellaropoulos, (2004) evaluated the environmental impact of waste wood co-utilization for energy production through life cycle assessment (LCA) by assessing the energy and materials consumption in relation to the amount of waste released into the environment during the life cycle of the system. The article was published in the ‘Energy’ journal in 2004. According to the authors, wood has specific characteristics such as high strength, low specific weight, good insulation and abundant availability. In Greece, the extensive use of wood results in residual waste wood in amounts of 1,070,000 m3/year. This is equivalent to 288,000 tons of oil (SKodras et al., 2004).
The co-utilization of waste wood with coal for energy production can help in reducing CO2 emissions while reducing the demand for fossil fuel and minimizing the problem of waste disposal. However, the co-combustion of waste wood may result in environmental problems, as it releases dioxins and furans (PCDD/F) in to the atmosphere. Moreover, various metal elements, such as Zn, Mn, Cd, and Ni, present in the waste wood may be released in gas phase due to combustion at high temperatures, thereby enhancing the formation of PCDD/F. To address these issues in terms of environmental advantages and disadvantages of using waste wood for co-combustion processes, the authors performed a life cycle assessment of related materials and the environmental outcomes from their use (SKodras et al., 2004).
The authors used the industrial boiler at a fiberboards (MDF) producing industry, PINDOS SA, situated in North-Western Greece for evaluating co-combustion processes. The boiler had two compartments with the first compartment containing a moving grate that covered nearly 50-60% of the total thermal input. Primary air and secondary air were distributed below and above the moving grate and the fuel supply, respectively. The second compartment included a multi-fuel combustion chamber containing saw dust, heavy oil, diesel or heavy oil combined with saw dust (SKodras et al., 2004).
Greek lignite, natural wood, MDF residues and power poles were employed for the co-combustion examination. Proximate, ultimate and ash analyses of fuels and assessment of the co-firing behavior were carried out, while monitoring periodically for oxygen concentration, and CO, SO2, NO emissions. Isokinetic sampling of the gas was carried out for PCDD/F followed by measurement with high resolution gas chromatograph-high resolution mass spectrometer. Ash samples were also collected to analyze unburned carbon and heavy metals. The resultant environmental impact from the process was evaluated using LCA formulated by the Society of Environmental Toxicology and Chemistry (SETAC). The LCA contains four steps, namely goal and scope definition, inventory analysis, impact assessment, and improvement analysis. Likewise, the environmental impact assessment by SETAC approach comprises four steps- classification, characterization, normalization and evaluation that are related to three parameters, namely the environmental protection, health safe, and resource depletion. By following these steps, the global environmental impact could be evaluated (SKodras et al., 2004).
Various fuel blends using waste wood and lignite were used for evaluation tests using LCA. The results showed that all waste wood blends proved to be good fuels, with stable combustion and emissions of CO, SO2, NO below the legislative limits. PCDD/Fs emission levels for all fuel blends varied between 3 and 97 pg/N m3, but below the legislative limit value of 100 pg/N m3. The majority of PCDD/Fs were found on the soot particles with only a small portion seen in the gas form. However, efforts to correlate of fuel blend composition and the percentage of wood in the fuel with the measured values of toxic emissions were unsuccessful. The concentration of heavy metals in the flues gases, except for zinc (1.4 mg/N m3), were found to be lower than the legislative limit value of 0.5 mg/N m3 (SKodras et al., 2004).
Coming to the environmental impact, it was found that the co-combustion of the waste wood with lignite in the existing industrial boiler resulted in decreased environmental effects. To be precise, the negative CO2 emissions during the period of wood growth accounted for low GHG emissions. Resource depletion and potential health impacts were also minimized, favoring co-combustion of waste wood with lignite using the available technology as a means of environmentally sound energy generation (SKodras et al., 2004).
Research Methodology
The methodology process of this study entails the collection, organization and integration of the collected data. Data collection will be the most important step in the success of this paper since it will lead to viable and credible findings. This study will be based on the research on several issues relating to the conversion of waste into energy. The study will also draw information and data from previous research conducted in the field of waste to energy applications. Though, the primary data through sponsored study by contacting various subjects through personal interviews and questionnaires will be ideal for the sake of hands on or firsthand experience, due to paucity of time and limited scope of this opportunity, the secondary data will be relied upon for the study. Nevertheless, this type of study as many scholars do is no less reliable as they are invariably supported by their own primary data. In addition case study on the chosen organization of ‘Polysius’ will be presented as a part of the study.
Review of Secondary Data and Analysis
Secondary research consists of the analysis of information and data gathered previously by other people like researchers, institutions and other non-governmental organizations. The data are usually collected for some other purposes other than one which is being presently attempted or it may help both the collection of data for both the studies (Cnossen). When undertaken with proper care and diligence secondary research can prove to be a cost-effective method in gaining better understanding of the specific issue being studied and conducting assessments of issues that do not need collection of primary data. The main advantage of secondary data is that it provides the basis for designing the primary research and often it is possible to compare the results of the primary research with secondary research results (Novak)
Research Design and Purpose
Data analysis and review in the secondary research method involves the collection and analysis of wide range of information. In order to conduct an efficient research it is important that a statement of purpose is developed first and a detailed definition of the purpose of the research is arrived at. It is also necessary that a proper research design is evolved. The statement of purpose is to have a clear understanding of the reasons for collecting the kind of data and the type of data the researcher wants to collect and analyze. This will help the researcher to stay focused on the topic under study and prevent from becoming overwhelmed with large volume of data. Research design can be defined as a step-by-step plan which shows the researcher the direction of data collection and analysis. The secondary data review normally involves designing the outline of what the researcher wants to study, the format of final report, list of type of data and a list of data sources that may be used for collection of data.
Sources of Secondary Data
There are different sources from which secondary data may be collected for the purpose of conducting any social research. The sources include:
- official statistics – include the official statistics collected by government departments and various other agencies, trade associations, information bureaus and other institutions like World Bank and International Monetary Fund (IMF). These statistics are particularly useful for the researchers since these data can be obtained more easily and comprehensive source of information that extends to longer periods of time. It is important that the official statistics collected as a part of the official statistics need to be verified for accuracy and reliability because according to Gill (1993) the official statistics are “characterized by unreliability, data gaps, over-aggregation, inaccuracies, mutual inconsistencies, and lack of timely reporting”.
- technical reports – these reports represent the research works previously carried out. They are made out to provide the results of the research to different research institutions, government and other interested research scholars. The report may be generated either out of a research already completed or from a research that is presently ongoing,
- scholarly journals – these sources generally contain reports generated out of original research or results of experiments conducted by scholarly people in the field. Articles in the secondary journals are subjected to a peer review where other knowledgeable people will make a critical analysis of the contents of the journal articles for the qualities of “accuracy, originality and relevance”.
- Literature Review Articles – review of literature gather and review articles of original research that deals with the topic under study. The reviews are normally presented by scholars and academics in the field of study and the reviews represent the overviews written for the first time on the topic. The review articles normally contain the list and details of all relevant publications and articles which formed the basis of review.
- Trade Journals and Review Books – While trade journals provide the practical information on the field under study the reference books form the secondary source where the researcher can find specific facts or summary of a topic discussed in detail. Handbooks, manuals, encyclopedias, and dictionaries are considered reference books ((University of Cincinnati; Pritchard and Scott)
Merits and Demerits of Secondary Sources
The main advantage of secondary data analysis is that it can be carried out more quickly as compared to the formal primary data collection and analysis procedure. If the quality of the secondary data available is relatively better the researchers can make best use of the secondary data rather than spending valuable time on the collection of primary data which will save a considerable effort on the part of the researcher. Using secondary data comparatively involves lower cost of gathering. However there is the element of variations in the collection of data between different researchers, countries and settings which needs to be considered while making use of the secondary data for analysis as the different methods may impair the comparability. Based on the level of data disaggregation the secondary data analysis can lead to a trend analysis to monitor changes in the values of attributes over a period of time. Secondary data can often be used to complement the primary data collection and thus will prove to save considerable time of the researcher in collecting the primary data. Secondary data facilitates the work of a researcher who lacks training in the collection and analysis of primary data (Beaulieu).
The main demerit of secondary sources of data is that they are considered as imperfect in nature as compared with primary data. Since there will be differences in the settings and purposes for which the data were collected a proper interpretation and analysis is required to understand the data presented and to use them for other purposes precisely. It is also necessary to apply selectivity without which there will be overwhelming volume of data that will impact the work of the researcher. It may be difficult to determine to judge the quality of some data which are being considered. Secondary data sources often conflict with each other. The original purpose for which the data was collected may bias the purpose of the secondary researcher.
Justification of the Secondary Research for Current Study
Though the gathering and analysis of primary data would add value to the reliability of the results of the research, in the chosen topic of study of the conversion of waste to energy, there are no enough opportunities within the time and reach of the researcher to resort to research methods of interviews or survey through questionnaires, since the firm involved in the study was fairly large and the executives of this large organization may not be easily be contacted for interview. It is also not possible to follow them for responding to the survey questionnaire as in their busy schedule they may not have time to devote to supplement this research. Moreover since the study proposed to derive the information and data for accomplishing the objectives of this research to be obtained from the published journal articles and research reports the secondary data become more reliable to be used for the purpose of the research. Since there is an abundance of literature on the subject and most of them can be considered as reliable sources the research has drawn the necessary information and data from the secondary sources. Therefore the secondary research has been considered suitable for completing the study.
Summary
This chapter presented a review of the secondary research method. It is observed that secondary data is found to be a valuable source of information for gaining knowledge and an in-depth idea on the broad range of issues and phenomenon. It is also found, though secondary research cannot replace primary research it is found to be useful in aiding the primary data collection. In view of the difficulties involved in using primary data collection methods and since the reliability of secondary sources of data is more it is justified to use secondary research for accomplishing the objectives of this study.
Bibliography
Barr, S., Gilg, A., & Ford, N. (2005). Defining the multi-dimensional aspects of household waste management. Resources, Conservation and Recycling , 45, 172-192.
Bebar, L., Martinak, P., Hajek, J., stehlik, P., Hajny, Z., & oral, J. (2002). Waste to energy in the field of thermal processing of waste. Applied Thermal Engineering , 22, 897-909.
Boran, J., Houdkova, L., Uckaj, V., & stehlik, P. (2008). Utilization of Energy from Thermal Treatment of Sludge. Management of Environmental Quality: An International Journal , 19 (4), 433-443.
Dajnak, D., & Lockwood, F. C. (2000). Use of Thermal Energy from Waste for Seawater Desalination. Deslianation , 130, 137-146.
Holmgren, K., & Henning, D. (2004). Comparison between Material and Energy Recovery of Municipal Waste from an Energy perspective: A Study of Two Swedish Municipalities. Resources, Conservation and Recycling , 43, 51-73.
Jain, A. K. (2007). Sustainable Development and Waste Management. Web.
Kollikkathara, N., Feng, H., & Stern, E. (2009). A Purview of Waste Management; Special Emphasis on USA. Waste Management , 29, 974-985.
Louis, G. E. (2004). A Historical context of municipal solid waste management in the United States. Waste Management and Research , 22, 306-322.
Phan, A. N., Ryu, C., Sharifi, V. N., & Swithenbank, J. (2008). Characterizatio of Slow Pyrolysis: Products from Segregated Wastes for Energy Production. Journal of Analytical and Applied Pyrolysis , 81, 65-71.
Porteous, A. (1998). Energy from Waste: A Whooly Acceptable Waste-Mamagement Solution. Applied Energy , 58 (4), 177-208.
Rulkens, W. (2008). Sewage Slides as a giomass resource for the production of energy: Overview and assessment of the various options. Energy & Fuels , 22, 9-15.
Skodras, G., Grammelis, P., Kakaras, E., & Sakellaropoulos, G. P. (2004). Evaluation of the Environmental Impact of Waste Wood Co-Utilization for Energy Producion. Energy , 29, 2181-2193.
SourcesofWaste. (2002). UN Atlas of the Oceans. Web.
Swihenbank, J., Nasserazadh, V., Wasanakorn, A., Lee, P. H., & Swithenbank, C. (2000). Futrure Integrated Waste, Energy, and Pollution Management (WEP) Systems Exploit Pyrotechnology. Journal of the Institute of Chemical Engineers , 78 (B), 383-398.
WasteWater. (1997). Pipeline. Web.