Introduction
Scientific experiment refers to a process of demonstrating a fact or a truth in the physical world. The experimental scientific method is a rational and logical order of steps followed to come up with conclusions. Gauch (2003).
Problem statement
How does acid rain affect the growth in height (cm) of bean plants?
Hypothesis
Plants watered with a mixture of water, and acid solution grows slowly than plants watered with distilled rainwater.
Relevance of the question
Many countries around the world have undergone the industrialization process. Many industries produce chemicals in the environment. Acid rain refers to the side effect of burning fossil fuels such as petroleum and coal. These sources produce nitrogen oxide or sulfur dioxide in the air when released. This leads to saturation of the earth’s atmosphere with large amounts of nitric acid (HNO3) and sulfuric acid (H2SO4). When snow or rain reaches the ground through a polluted atmosphere, plants are damaged, White (2009).
Literature review
According to Driscoll (2009), Acid rain is a complex atmospheric and chemical phenomenon that occurs because of emissions of air pollutants like Sulfur Dioxide (SO2) and nitrogen oxides (NO2). They react with the atmosphere to form acidic compounds such as sulfuric acid and nitric acid. The acid deposition includes dry and wet forms. The compounds return to earth in wet form as snow, fog, or as acid rain. The acids are chemically transformed to SO2 or nitrogen and sulfur salts. In these forms, they are deposited in dry form, which causes the same damage as when deposited in dry foam. SO2 and NO2 go through chemical reactions and they are converted to sulfuric acid (H2 SO4) or nitric acid (HNO3). The reactions that occur are as follows:
- 2SO4 + O2 = 2SO3
Sulfur trioxide then reacts with water to form sulfuric acid;
- SO3 + H2O = H2SO4
Nitrogen dioxide and water reacts to form nitrous acid and nitric acid;
- 2NO2 + H2O = HNO3 + HNO2
Normal rain has a PH of 5-6 because of atmospheric reactions involving carbon dioxide. Therefore, acid rain has a PH of less than 5 and it harms the growth of plants when it falls on them.
Sources of acid rain
According to (Burns 2004), acid rain is because of two processes. Hydrochloric acid, caused by secondary pollutants from oxidation of SO2 and NO2 gases, is released into the atmosphere. The process of converting these gases into acids takes several days. They are transferred over long distances during this period. The longer the gases stay in the atmosphere, the more they are converted into an acidic component. Burns argues emissions of SO2 are responsible for 60-70% of acid rain that occurs worldwide. 90% of sulfur is caused by human activities while 95% of NO2 is caused by human activities. Coalfields, natural gas processing plants and ore smelting forms the major source of these gases Borer (2009).
Effects of acid rain
According to Bailey (2005), acid deposition causes environmental damages, acidification of lakes and rivers, impairment of air quality, and health problems. Acid rain has stressed trees and a decline in the growth of plants. Acid rain leaches elements from plant foliage and makes them susceptible to freezing. Acid deposition alters soil by accelerating the leaching of base cations and increasing the concentration of dissolved inorganic aluminum in the soil. It has also affected water in rivers and lakes by increasing acidity levels thus aquatic plants cannot grow.
Experimental design
The experiment aims at growing 180 bean plants that will help to test the effect of acid rain on the appearance and growth of the specimen plants. The researcher aims at collecting and testing the rainwater for PH levels. Ninety of the plants will be weeded out and the remaining specimen will be used to build a data table that will be important in the discussion of the results of acid rain on the environment. The remaining 90 plants are divided into five different groups of 18 plants each. Group 1 will be the control group and it will be watered with distilled water with a PH of 7.0. Group 2 will have a mixture of sulfuric acid with a PH 0f -1, group 3 will have sulfuric acid with a PH of 1, group 4 will have butter solution with a PH of 4, and group five have Nitric acid with a PH of 5. The groups will be watered twice per week in the evening with their specific solutions. The height of the plants will be measured twice per week (Monday and Friday) and by one person to ensure accuracy. Rulers will be used for measurements recorded in centimeters. Photographs were taken using cameras. The result of the groups will be compared with the control group. The plants will also receive fertilizer to provide them with nutrients. The experiment took 6 weeks. Once we calculate the acid, we add it to the distilled water to get the desired PH, the solutions will be tested using PH test strips.
Data collection
Data collection is an essential part of the research study. The methods used are observation, recording, and photography. This involves physical observation of the plant as it grows, measuring, and recording the height using rulers. Photographs will show the increase in height of the plant.
Photograph of the bean plant in various stages of development

Justification of tools, technologies, and methods
The usage of a ruler is justified because it has measurements in centimeters, which are our units of measurement. Rulers are cheap to obtain and are locally available. The use of the camera is justified in that it will provide a chronology of the growth of the plant through pictures from seedling to maturity. The use of lab coats and groves is justified to protect the researchers from dirt and diseases. The observation method is the best to research a non-regulated environment.
Independent and dependent variables
The dependent variable in the experiment will be the height of the bean plant. This is because the height of the plant will depend on the amount of acid water mixture used. The independent variable will be the mixture of the acid and water because this will remain constant throughout the experiment. Fertilizer will be an extraneous variable because the plants will show signs of stunted growth in its absence due to lack of nutrients.
Control variable for the experiment
The independent control variable will be distilled water while the dependent control variable will be the plants in distilled water Legge, (2009).
Results
The high levels of acidity affected the growth in height of the bean plant. The graph below shows the average heights of the bean plant. An obvious inverse relationship was observed between the amount of acid activity and the height of the bean plant.
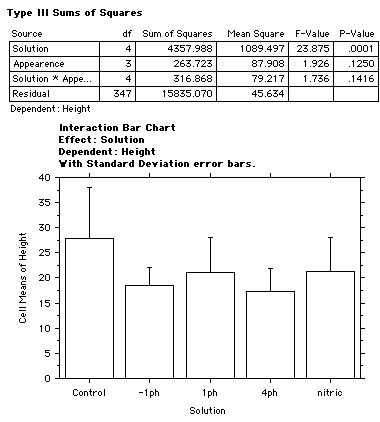
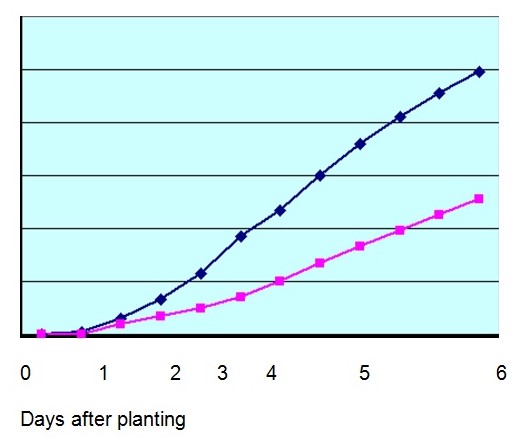
Figure 2 shows the normal growth of the plant in a control group.
Plants with high acidity deteriorated faster as shown below:

Replication of the experiment
Any experiments conducted must be repeatable. Replication will allow the researcher to make statistical tests useful in evaluating observed features. Replication to be done by increasing the number of observations will strengthen the information obtained from the observation. Replication will also include variations not included in the experiment. It will include a control where all the variables are constant except for the variable under investigation.
Discussion and Conclusion
The results confirm the hypothesis that acid water harms the growth of plants. The experiment shows that plants grew best in PH levels of 7.0 and that plants are not acid tolerant. This is because acid destroys microorganisms that thrive in the soil. Acid affects the number of nutrients available for plants in the soil. In a solution of acid and water, plants had stunted growth and they eventually died while the plants in distilled water had normal growth. Industries should control the emission of industrial gases like Sulfur dioxide and Nitric acid that led to acid rain. Countries should put in regulations to control the emission of gases.
References
Bailey, M. J. (2006). Microbial Ecology of Aerial Plant Surfaces. New York: CABI.
Borer, H. (2009). The Normal Course of Events. London: Oxford University Press.
Burns, C. M. (2004). Ecological Interface Design. New York: CRC Press.
Driscoll, J. (2009). Environmental and forest biology. Chicago: Springfield Publishers.
Gauch, H. G. ( 2003). Scientific Method in Practice. London: Cambridge University Press.
Legge, A. H. (2009). Air Quality and Ecological Impacts: Relating Sources to Effects. London: Elsevier.
White, J. C. (2009). Acid Rain: The Relationship Between Sources and Receptors. New York: Springer.