Results
Section 1
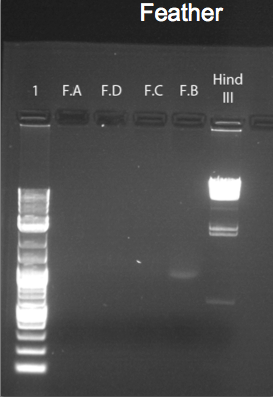
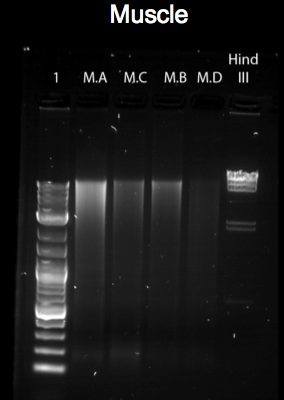
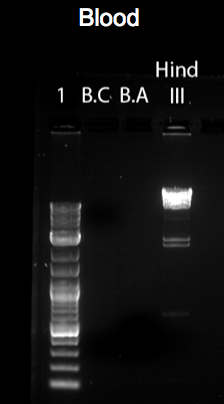
The figures above represent gel-electrophoresis test results of a test performed from blood, muscle and feather of a Gallus gallus DNA samples. The figures also highlight the exact measurements of DNA concentration using samples from a Nano-drop machine. Notably, the figures illustrate clear differences in the amount of DNA collected from the samples. For instance, no band appears on the gel of the samples of feather and blood in figures 3 and 1 above. This shows that the DNA extracted from the sample was minimal. The difference can be clearly noted in the bands formed in figure 2. In this case, the muscle DNA sample extraction was more than the other samples. The table below clearly provides DNA concentrations in ng/uL of the tissue types used.
Table 1: DNA concentration of different tissue types
The use of a Nano-drop machine in conducting this test was to determine the concentration of DNA of the used samples based on their absorbance. This was achieved by measuring the absorption of light by the samples. In that case, the concentration of DNA in a sample was found to be high when surplus amount of light was absorbed. The table above explains the differences of DNA concentration of the different sets of tissues used. From the amount of light absorbed, DNA extraction from the muscle tissue appeared high because it recorded a mean of 35.8. The other samples showed DNA mean concentration for blood at 10.7 and that of a feather at 5.5. To clearly show the difference in the results, the study used a bar chart to point out the mean of DNA concentrations of the samples. See below a bar chart reflecting the different concentrations of the tissues.
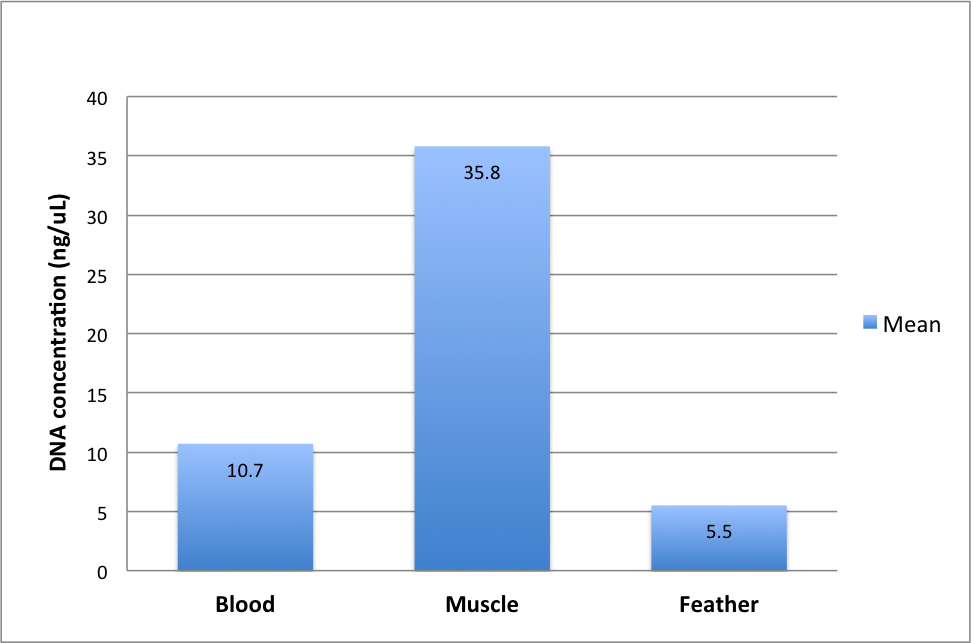
In the bar chart above, it is vivid that the quality of DNA of the tissue types used greatly varied. Besides, it can also be noted that with a low DNA concentration, a sample can still offer good concentration quality without regard to the quantity or quality of DNA extracted. It is imperative to note that the purity of DNA has can be assessed when the absorbance ratio of the samples used are between 260nm and 280nm. The table below shows how the purity of the samples used has been determined by the absorbance ratio at 260/280.
Table 2 Ratio of absorbance at 260/280 in different tissues
The table above carefully explains the quality of the different samples used. One remarkable observation on the table is the representation of blood tissue sample that shows very poor DNA quality of between 1.32 and 1.36. However, samples of muscle and feather have a high DNA quality of 2.17 and 1.70 in feather and muscle B respectively. This shows that to effectively determine the quality of DNA, it is important to rely on a tissue sample and not a tissue type.
Section 2
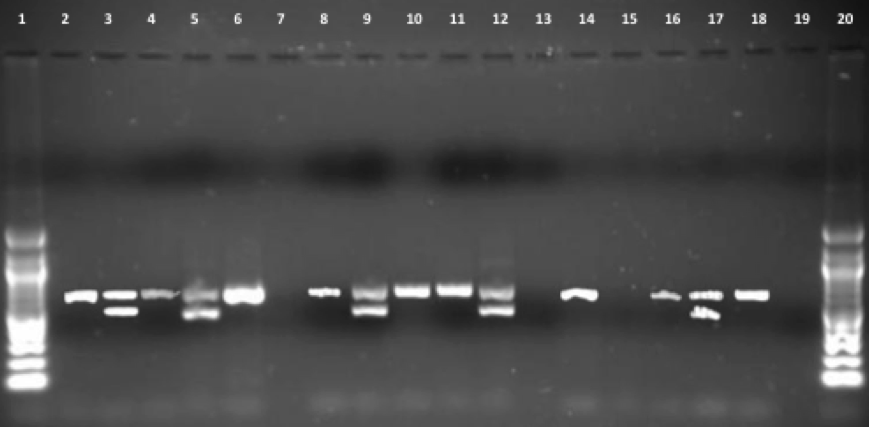
Figure 5 above presents result from Gallus gallus blood, muscle and feather DNA extraction using PCR of the gel electrophoresis. The outcomes show observable sexual characteristics of the blood band for males and females. Blood A in the third band in the figure above correlates with the two bands representing female positive control. On the other hand, the Z chromosome in blood B in lanes 2 and 4 represents a male.
Besides, lanes 8 and 10 on feathers show observable sex features. Sample feather B is for a male character. This can be noted by the one band formed and well presented by a Z chromosome. This is quite the opposite in lane 9 which has two bands, a representation that feather A is of a female sample.
The amplification of bands in the muscle samples results is poor and not clearly defined. This makes the determination of the sex a bit problematic. When you observe the 15th lane, it is clear that there are no bands representing muscle sample A. However, there is a band of muscle B at lanes “14” and “16”. This significantly helps to determine the sex of the sample as male as the band is well located in gene Z.
In addition, the study conducted a comparison of the samples using a 100bp ladder. It was vivid that genes Z and W manifested major differences in their base pairs. One such significant difference was that the W gene pair had a base of approximately 600 while the Z gene had a base pair of 700. To determine the final result of the tests, the study used six samples with two samples derived from each of the three tissues. At the end of the test, the results showed that the tissues had one unknown sex, three males and two females. Muscle A as observed in lane 5 had no bands at all and was represented by muscle A. The analysis showed massive amplification in the diverse tissue types with some similar to those in lanes 2 and 3 showing perfect amplification while those in lanes 15 and 16 showing faded and imperfect bands.
Discussions
The practical aim of this study was to use the different tissue samples to determine the sex of the Gallus gallus species. In the study, table two clearly showed the differences in DNA concentrations. Muscle tissue B as noted in the table yielded the highest concentration of DNA followed by the blood tissue. The expectation of the study was that blood, being made of components such as the nucleus and huge amounts of white blood cells would have more DNA. However, this was not the case from the sample because it gave less amount of DNA compared to the muscle tissue. The least DNA concentration came from the feather sample. This was caused by the fact that it was only possible to collect DNA for the tip of the feather.
The study had several limitations such as the presence of imperfect bands formed despite the availability of high DNA concentration in the samples. This was observed in the muscle and blood samples. The problem could have been brought about by errors in sampling and using instruments. This could have made the bands to form poorly. Besides, the storage of the samples used might have contributed to the problem. It is believed that poor and improper storage of samples without consideration of the effects of temperature on DNA degradation was another factor that gave rise to undesired results.
Besides, an effective analysis of a sample requires determining whether or not it is invasive. A non-invasive sample offers technicalities in determining results of the tissue used. In this study, it appears that there was a non-destructive blood collection, a consideration that made it impossible to determine high DNA concentration of the samples. One important factor to note in collection and use of a muscle sample is that a destructive sample negatively impacts on the results because it lowers the DNA yield.
In recent years, determining the sex of a bird was easy because observable features such as the gonads, monomorphic traits, brooding patch as well as behavioural observations were largely ignored. However, this practise was only effective when conducted during breeding seasons. The use of gel and PCR to determine the sexing primers of birds has made it possible to know the sex of a bird. It is imperative to note that the genetically male birds are ZZ homozygous while female ones are heterozygous ZW. The existence of molecular identification of sex via CHD using gel has made discovery of gene Z and W possible. The use of sex primers therefore enables the tests to be conducted without waiting for breeding seasons to come. The only thing need is PCR and gel electrophoresis of live tissues containing nucleus of a bird or a species sample.