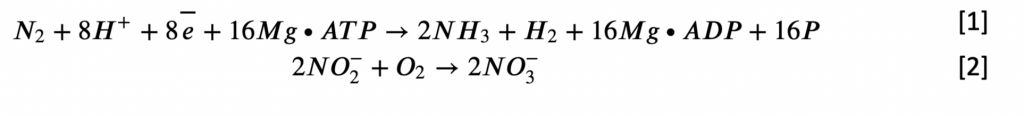Virtually every chemical element on the Periodic Table is vital for sustaining life, but some of them, and nitrogen, in particular, are more important. Nitrogen is involved in a grandiose number of natural elements of the biosphere, including plant and animal proteins, air, dead organic matter, and soil mineral nutrients. Such a variety of nitrogen functions naturally gives rise to a diversity of living organisms that have the biological capacity to fix molecular nitrogen. For instance, biological nitrogen fixation is accomplished through capture by plant leaves or nitrogen-fixing soil bacteria and blue-green algae. Each of the mentioned organisms transforms molecular nitrogen into organic compounds as a result of metabolic processes. In turn, ammonifying bacteria, also known as decomposition ones, create ammonium and nitrite compounds from the residues, which are used by nitrifying prokaryotes to produce energy.
The recycling produces large amounts of nitrates, which are reused as food by the plant’s roots, along with ammonium salts. Finally, the soil nitrates are reduced by denitrifying bacteria to molecular nitrogen that enters the atmosphere: the cycle’s biological foundation is complete. In the nitrogen cycle considered, it has been repeatedly stated that micro and macroorganisms are capable of transforming substances containing this element. It follows that the mechanisms of biological fixation must be adaptive for each individual taxon. For example, there is the extremely important anaerobic model of atmospheric nitrogen fixation, which proceeds only in the absence of oxygen access.
The nitrogen molecule is trapped by soil bacteria and then activated by the enzyme nitrogenase. Using the complex protein ferredoxin as a cofactor, nitrogenase reduces nitrogen to ammonia, as shown in Equation (1). Another vital model of nitrogen fixation is aerobic, which, conversely, requires the presence of oxygen. In particular, proteins and other organic nitrogen-containing substances are the products of organisms, so they are abundant in soil and terrestrial environments. Bacteria, particularly the Azotobacter genus, hydrolytically break down complex substances with the help of proteases, and under aerobic conditions, the decomposition of molecules goes all the way to the final products. Moreover, an alternative process is nitrification, whereby nitrites interact with molecular oxygen to form nitrates, a simplified equation of which is shown in (2).