Introduction
A multi-component reaction (MCRs) is a combination of chemistry and molecular biology in solving the emerging issues in the medical industry. The reaction is not only environmental friendly but also cheap thus its frequent application in the manufacture of triazine derivatives. Currently, scientists are applying MCR technique in the synthesis of drugs from different chemical elements like triazines. Triazines are chemical, unsaturated N-heterocycles compounds. The heterocyclic ring has three carbon atoms and three nitrogen atoms. Triazines have three isomeric forms α (1, 2, 3 triazine), β (1, 2, 4 triazine) and γ (1, 3, 5 triazine). The structure of the three isomers differ at the location of the nitrogen atom in the ring thus culminating into 1, 2, 3 triazine, 1, 2, 4 triazine, and 1, 3, 5 triazine. The chemical synthesis of triazines differs from one isomer to the other. The laboratory synthesis of the triazines is through multicomponent reactions whereby, more than three essential chemical elements undergo chemical reaction to generate a new compound containing properties of the individual elements/reagents.. Therefore, there are a number of methods used in generating triazines among them, trimerization, Bamberger reaction, and Pinner reaction (Hansjuergen and Grundmann 2450). Additionally, 1-azadienes are applicable in the synthesis of N-heterocycles like triazinane diones, which is a derivative of triazine. On the other hand, beta-secretase (BACE) is an aspartate protein that initiates the development of Alzheimer’s disease in the human body. Thus, BACE-1 inhibitors are drugs developed to stop the pathogenesis of the disease but the Bace1 proteins turn out to be complicated. Triazines can apply in medicinal chemistry especially as BACE-1 inhibitors because of the structural similarity with other BACE-1 inhibitors. Thus, scientists are trying to get alternatives and analogs of triazine have proven to be promising due to the structural similarity with other BACE-1 inhibitors.
Ways of making triazines
The synthesis of triazine diones occur commonly through carbon-based reactions but the use of isocyanates, nitriles, phosponates and aldehydes still remains unexploited. However, the common method in the synthesis of triazines is by using multicomponent reaction whereby all the reagents are in one container and the all the necessary chemical processes applied (Groenendal et al. 720). Using a single process there is generation of the main compound and with subsequent reactions, there is production of the final element. Artificially, Triazanone, a derivative of triazine is one of the compounds chemically synthesized through MCR. According to Yakup, triazines can exist in different forms like mono, di, and trinitro among others (Para.1). Therefore, there are three methods used to synthesize Triazines depending on the derivative or final product. For instance, For instance, generation of β-triazines from 2-azidocyclopropenes, which is through heat application and the synthetic derivation of α-triazines, is through combination of1, 2-dicarbonyl compounds, and amidrazones). The synthetic production of γ-triazines is through trimerization of cyanimide (Dryer, Blume, MonikaJorg, Neumann-Rodekirch para.2). Artificially, 1-azaidienes applies in the production of nitrogen heterocycles like triazines (Erickson and Wiley 3). Due to the structural diversity and complexity, 1-azadienes can react as nucleophiles, electrophiles and heterodynes among many other forms. Thus, the artificial production of triazinane diones especially for use in the pharmaceutical industries relies heavily on 1-azaidienes (Simons, and Saxton 79). Moreover, through MCR there can be production of 1-azaidienes on large scale, which eventually processes analogs of triazines. In the Bamberger reaction, chemical reagents undergo three steps to produce commercial triazine (Hassner and Stumer 5).Aniline reacts with sodium nitrate forming aryl diazonium, which disintegrates into an intermediate product (azo). According to Paravidino et al., hydrochloric acid and sulfuric acid are biocatalysts, which initiate the disintegration of the reagents and eventually the intermediate into benzotriazine (5370).
In Pinner method combinations of aryl amidines plus phosgene condense together forming 2-hydroxy-4, 6-diaryl-sym-triazines (Vugts 7178). The final product culminates from unstable intermediate called bismidyl urea. In trimerization, Cyanogen chloride is unstable compound with the ability to react continuously until it forms polymers a property, which qualifies it to form trimmers (Hansjuergen, and Grundman 2449). Furthermore, trimerization of amidines also yields symmetrical triazines as the products and when 2-azidocyclopropene undergoes heating, there is chemical production of α-triazines.
According to Shie and Fang, the synthesis of triazines through multicomponent reaction can be through heating of primary alcohols combined with aldehydes at the temperature of 80 degrees Celsius (3142). The chemical process involves reaction of many primary alcohols using ammonia and iodine in the presence of radioactive particles (Zhu and Bienayme 80). Finally, through addition of dicyandiamide plus sodium azide to the intermediate product, which is nitriles, triazines become the final products. In the figures below, adopted from Shie and Fang (3141), it is evident that heat application is an important element during the production of triazines and some of its derivatives. Therefore, from the chemical process below, temperature or heat application is the crucial element in the laboratory synthesis of triazines. The discovery of microwave radiation has led to the improvement of triazine synthesis, as it is the case below.
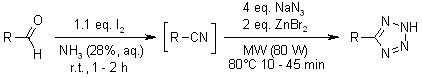
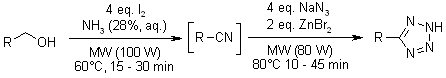
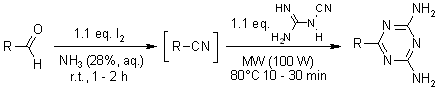
Structures of BACE-1 inhibitors
Beta secretase is a protease enzyme responsible for the emergence of Alzheimer’s disease in old age (Huang et al, 5437). To inhibit the pathogenesis of the Alzheimer’s disease, scientists have invented Bace1 inhibitors thus curbing the disease. The inhibitors dock or lodge into the enzyme disabling it from catalyzing the progression of the disease (Zhaoning et al. 951). There are different types of Bace1 inhibitors with chemical structures similar to triazines in tetrahedral forms (Yong 3).The common structure of the Bace1 inhibitors has two benzene-closed rings and three open rings with seven nitrogen atoms attached. The structure also has both nitrogen and hydrogen bonds connecting it. Chemically, hydrogen bonds are weak and therefore may lead a compound to have a poor structure. The Bace1 inhibitors are not only unsaturated, but also consist of two hydrogen atoms attached to the nitrogen atoms. The active compound, which is similar to the Bace1 enzyme, differs slightly depending on the reagents or chemicals used. Through the application of the lock and key hypothesis, the Bace1inhibitors, which have a similar structure to the active Bace1 enzyme, stop the pathogenesis of the disease (Leach 25). The inhibitors block the active sites of the enzyme inactivating it and eventually halting the disease (Xu et al. “Identification of a sub-micromolar” 5763). Thusly, the structural compositions of Bace1 inhibitors like KIM-358 and KIM-370 either improve its potency against the Bace1 enzyme or decrease the activity levels.
The use of triazines as Bace1 inhibitors and the relationship between the two inhibitors
Laboratory studies have shown that thioisourea 3 occupies the active site of the inhibitor thus; a lead compound is applicable in the development of the inhibitor. Though originating from lead (nonpeptide) the analogs/derivatives of triazines may apply as inhibitors due to possession of good properties as protein-ligand components. A preliminary study on mice has shown that a compound of lead composed of 2-amino-dihydropyrimidinone ring has a biphenyl compound at carbon number 6 that acted as a precursor for the development of the inhibitor. Through coupling (Suzuki method) and use of 3-bromobenzaldehyde there is formation of a ring (aromatic). Additionally, hydrogenation of the aromatic ring converts the urea into guanidine, which is the essential component in the construction of the triazanone diones as a Bace1 inhibitor. The structure of the triazanone diones draws similarity from the Bace1 inhibitors but the possession of a guanidine moiety makes the former to exhibit better properties like stability and efficient activity against the Bace1 enzyme.Triazines and other Bace1 inhibitors possess common benzene ring despite the fact that they originate from different materials or reagents during laboratory synthesis. For instance, when Huang et al. discovered other Bace1 inhibitors, the stable compounds (inhibitors) had similar structures to the triazine, which shared a triazine scaffold (5436). Chemically, triazine structure possesses an enclosed ring with three nitrogen atoms and three carbon atoms attached (Hinchliffe 17). Therefore, the skeleton structure of triazine is similar to all other Bace1 inhibitors. The chemical structure and composition of triazine compounds increases its potency as a Bace1 inhibitor. Furthermore, the compounds, which have triazine scaffold, are able to react in mammalian cells, which mean they have high cell permeability (Xu et al. “Molecular docking” 6205). In conclusion, it is recommendable to use triazines as a source of nonpepetides Bace1 inhibitors.
Unfortunately, the instability of the commonly used Bace1 inhibitors prompted scientists to search for an alternative and luckily, triazine and its other derivatives may solve the problem. Though not practiced in medicinal chemistry, Godemann et al. recommended the use of triazines or the derivatives as Bace1 inhibitors because the compounds are stable during chemical reactions (10743). Triazines may reduce both cases of emerging Alzheimer’s and stop the spread or the impact of the disease. During the laboratory experiment, the common inhibitors like KMI-358 and KMI-370 undergo isomerization to α-N-oxalyl-DAP derivatives, which is due to the instability nature of the inhibitors. Nevertheless, the addition of triazines or tetrazoles increases both the chemical stability and inhibition potency (Chadwick, Kellett, Hooper, Johnson, and Fishwick 6772). Therefore, triazine may not only apply as Bace1 inhibitor, but may also improve the quality of the existing inhibitors. Moreover, the high boiling temperature of about 114˚C and high melting point of 86˚C increase the chemical stability of the triazine structure and its derivatives. The high boiling point is due to the possession of strong nitrogen bonds, which increase the strength of the compound. The ability to improve the protein-ligand properties of the existing Bace1 inhibitors may lead to application of triazine as a bace1 inhibitor in medicinal industry.
The chemical and physical properties of triazines improve its chances to inhibit enzyme Bace1 protease. The s-triazine compound has high thermal stability and easier to dissolve in solvents like ether (Silvestri 356). The ability to dissolve easily makes it easier to purify triazines thus making it cheaper and readily available in pharmaceutical industries or medicinal chemistry. Additionally, the high volatile nature of the triazines allow easier purification using nitrogen or dry air (Smolin and Rapoport 30).The melting point changes insignificantly especially with the addition of other elements or atoms like nitrogen. Triazines also have high heat of combustion, which increases the level of stability against other chemical compounds. Therefore, the possession of unique properties increases the chances of triazine as a Bace1 inhibitor. With unique chemical and physical properties, both the chemical, pharmaceutical, and medicinal industries may recommend the use of triazine as either stabilizer of Bace1 inhibitors or as an inhibitor incases of Alzheimer’s disease. With advanced molecular chemistry, MCRs have seen the development of diverse libraries vital in solving many biologically issues especially in production of exceptional interactive patterns between “β-Site Amyloid Cleaving Enzyme (BACE) catalytic diad and a weak NMR screening hit.
Conclusion
In summary, the isomers of triazine have a similar skeletal compound consisting of an enclosed benzene ring with three nitrogen atoms and three hydrogen atoms attached. The formation of triazines may be through trimerization, condensation of different chemical elements or through a series of chemical reactions the Bamberger and the Pinner methodologies. The procedure of the chemical synthesis is in accordance with final product or isomer/derivative desired. Additionally, as regards to triazine synthesis, the process has greatly changed: from combinatorial synthesis to orthogonal safety catch methodology that has seen for the generation of novel analogs necessitating biological processes. The use of heat and radioactive particles is the most common method applied in chemical synthesis of triazines. Many research scientists recommend the use of triazine in medicine especially in the treatment of Alzheimer’s disease.
Works Cited
Chadwick, James, Kellett, Katherine, Hooper, Nigel, Johnson, Peter, and Fishwick, Colin. “Discovery of novel non-peptide inhibitors of BACE-1 using virtual high-throughput screening.” Bioorganic and medicinal chemistry letters 19.23 (2009): 6770-6774. Print
Dryer, Christian, Blume, Alfred, MonikaJorg, Bauer, Neumann-Rodekirch, Jens. Fourth International Electronic Conference on Synthetic Organic Chemistry (ECSOC-4), September 2000.
Erickson, John, and Wiley, Paul.The Chemistry of Heterocyclic Compounds. New York: Wiley, 1956.
Godemann, Robert, Madden, James, Kramers, Joachim, Myron, Smith, Ulrike Fritz, Hesterkamp, Thomas, Barker, John, Hoppner, Sabine, Hallett, David, Cesura, Andrea, Ebneth, Andreas, and Kemp, John. “Fragment-Based Discovery of BACE1 Inhibitors Using Functional Assays.” Biochemistry 48.45 (2009): 10743. Print.
Groenendal, Bas, Vugts, Danielle, Schmitz, Rob, Frans de Kanter, Eelco, Ruijter, Groen, Marinus, and Orru, Romano.” A Multicomponent Synthesis of Triazinane Diones.” Journal of Organic Chemistry 73.2 (2008): 719–722.
Hansjuergen, Schroeder, and, Grundmann, Christopher. “Triazines XIV. The Extension Of the Pinner Synthesis of Monohydroxy-s-triazines to the Aliphatic Series. 2, 4-Dimethyl-s-triazine.” Journal of American Chemistry Society 78.11(1956): 2447–2451. Print
Hassner, Alfred, and Stumer, Carol. Organic Synthesis Based on Name Reactions: Tetrahedron Organic Chemistry Series. Oxford: Pergamon. Hinchliffe, Allan. Modeling Molecular Structures. New York: Wiley, 1996.
Huang, Danzhi, Urs Lu¨ thi Kolb, Peter, Cecchini Marco, Barberis, Alcide, and Caflisch Amedeo. “In Silico Discovery of â-Secretase Inhibitors.” Journal of American society 128.16 (2006): 5436-5443. Print
Leach, Andrew. Molecular Modeling. England: Addison-Wesley/Longman, 1996. Paravidino, Monica, Bon Robin, Scheffelaar, Rachael, Vugts Danielle, Anass, Znabet, Schmitz, Rob, Frans de Kanter, Lutz, Martin, Spek, Anthony, Groen, Marinus, and Orru, Romano. “Diastereoselective Multicomponent Synthesis of Dihydropyridones with and Isocyanides Functionality.” Organic letters 8.23 (2006): 5369-5372. Print
Shie, Jiun, and Fang, Jim. “Microwave-Assisted One-Pot Tandem Reactions for Direct Conversion of Primary Alcohols and Aldehydes to Triazines and Tetrazoles in Aqueous Media.” Journal of Organic Chemistry 72.2 (2007): 3141-3144.
Silvestri, Romano. “Boom in the development of non-peptidic beta-secretase (BACE1) Inhibitors for the treatment of Alzheimer’s disease.” Medicinal Research Review 29.5 (2009): 295-338. Print.
Simons, John, and Saxton, Michael. “Benzoguanamine: s-Triazine, 2, 4-diamino-6- Phenyl.” Organic Syntheses 33.4 (1963): 78-80.
Smolin, Edwin, and Rapoport, Lorence. S-Triazines and Derivatives. New York: Interscience Publishers, 1967.
Vugts, Danielle, Manoe Koningstein, Rob Schmitz, Frans de Kanter, Marinus B. Groen, and Romano, Orru. “Multicomponent Synthesis of Dihydropyrimidines and Thiaz.” Chemistry European journal 12.2 (2006): 7178. Print
Xu, Weijun, Gang, Chen, Weiliang, Zhu, and Zhili, Zuo. “Molecular docking and structure–activity relationship studies on benzothiazole based non-peptidic BACE-1 inhibitors.” Bioorganic and medicinal chemistry letters 20.21 (2010): 6203-6207
Xu, Weijun, Gang, Chen, Weiliang, Zhu, and Zhili, Zuo. “Identification of a sub-micromolar, non-peptide inhibitor of β-secretase with low neural cytotoxicity Through in silico screening.” Bioorganic and medicinal chemistry letters 20.19 (2010): 5763-5766. Print
Yakup, Camur. A Computational study on nitrotriazine derivatives, 2008. Web.
Yong, Gong, Mark, Bausch, and Linhua, Wang. “Acylimine mediated n_n bond cleavage Of pyrazolidinediones and subsequent conversion to dihydropyrimidinediones and malonamides.” Tetrahedron letters 42.4 (2001): 1-4. Print
Zhaoning, Zhu, Zhong-Yue, Sun, Yuanzan, Ye, Johannes, Voigt, Corey Strickland, Smith, Elizabeth, Cumming, Jared, Wang, Lingyan, Wong, Jesse, Wang, Yu-Sen, Wyss, Daniel, Xia, Chen, Kuvelkar, Reshma, Kennedy, Matthew, Favreau, Leonard, Parker, Eric, McKittrick, Brian, Stamford, Andrew, Czarniecki, William, Michael, Greenlee, and John, Hunter. “Discovery of Cyclic Acylguanidines as Highly Potent and Selective β-Site Amyloid Cleaving Enzyme (BACE) Inhibitors: Part I; Inhibitor Design and Validation.” Journal of medicinal chemistry 53.4 (2010): 950-59. Print
Zhu, Jieping, and Bienayme, Hugues. Multicomponent reactions. New York: Wiley, 2005.